19. Chemical Thermodynamics
Spontaneous vs Nonspontaneous Reactions
Problem 13
Textbook Question
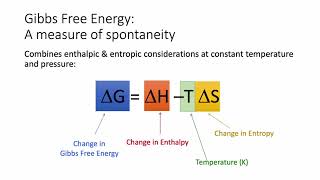
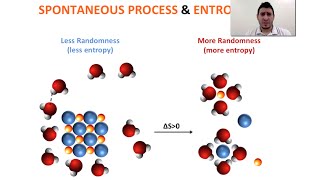
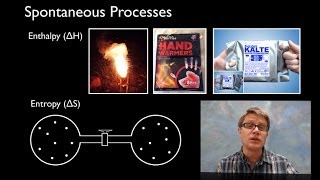
Textbook QuestionIndicate whether each statement is true or false. (a) A reaction that is spontaneous in one direction will be nonspontaneous in the reverse direction under the same reaction conditions. (b) All spontaneous processes are fast. (c) Most spontaneous processes are reversible. (d) An isothermal process is one in which the system loses no heat. (e) The maximum amount of work can be accomplished by an irreversible process rather than a reversible one.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
926
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 9 videos