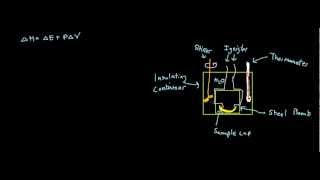
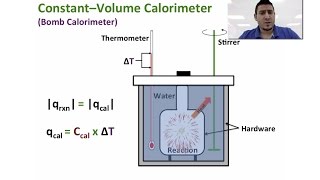
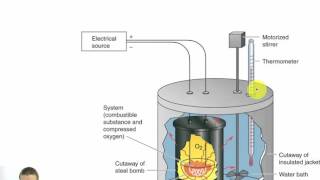
8. Thermochemistry
Constant-Volume Calorimetry
Problem 111
Textbook Question
Textbook QuestionFrom the following data for three prospective fuels, calculate which could provide the most energy per unit mass and per unit volume: Fuel Density at 101.3 kPa (g/cm3) Molar Enthalpyof Combustion (MJ/mol) Octane, C8H181l2 0.70 at 20 °C -5.53 Liquid Butane, C4H101l2 0.60 at -1 °C -2.88 Liquid hydrogen, H21l2 0.07 at -253 °C -0.29
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
7mPlay a video:
559
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos