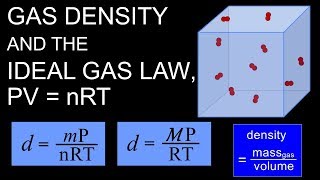
7. Gases
The Ideal Gas Law Derivations
Problem 109
Textbook Question
Textbook QuestionAn ideal gas at a pressure of 152 kPa is contained in a bulb of unknown volume. A stopcock is used to connect this bulb with a previously evacuated bulb that has a volume of 0.800 L as shown here. When the stopcock is opened, the gas expands into the empty bulb. If the temperature is held constant during this process and the final pressure is 92.66 kPa, what is the volume of the bulb that was originally filled with gas?

 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
727
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 12 videos