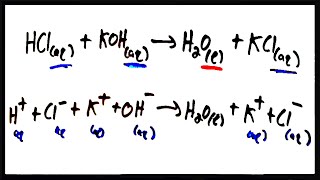6. Chemical Quantities & Aqueous Reactions
Molecular Equations
Problem 5
Textbook Question
Textbook QuestionYou are presented with a white solid and told that due to careless labeling it is not clear if the substance is barium chloride, lead chloride, or zinc chloride. When you transfer the solid to a beaker and add water, the solid dissolves to give a clear solution. Next an Na2SO41aq2 solution is added and a white precipitate forms. What is the identity of the unknown white solid?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
424
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 9 videos