16. Chemical Equilibrium
ICE Charts
Problem 146
Textbook Question
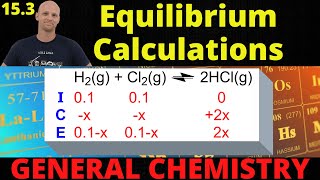
Textbook QuestionRefining petroleum involves cracking large hydrocar- bon molecules into smaller, more volatile pieces. A simple example of hydrocarbon cracking is the gas-phase thermal decomposition of butane to give ethane and ethylene: (c) A sample of butane having a pressure of 50 atm is heated at 500 °C in a closed container at constant volume. When equilibrium is reached, what percentage of the butane has been converted to ethane and ethylene? What is the total pressure at equilibrium?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
4mPlay a video:
315
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 12 videos