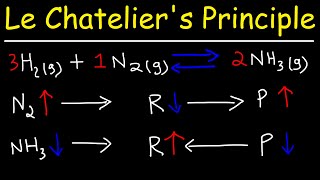
16. Chemical Equilibrium
Le Chatelier's Principle
Problem 129
Textbook Question
Textbook QuestionThe binding of oxygen by hemoglobin in the blood involves the equilibrium reaction: HbH+(aq) + O2(aq)HbO2(aq) + H+(aq) In this equation, Hb is hemoglobin. The pH of normal human blood is highly controlled within a range of 7.35 to 7.45. Given the above equilibrium, why is this important? What would happen to the oxygen-carrying capacity of hemoglobin if blood became too acidic (a dangerous condition known as acidosis)?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
0m:0sPlay a video:
10025
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 13 videos