24. Transition Metals and Coordination Compounds
Strong-Field vs Weak-Field Ligands
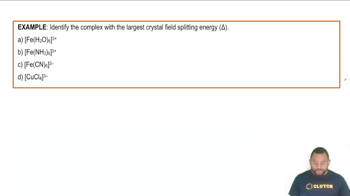
Problem 104
Textbook Question
Textbook QuestionThe [Ti(NCS)6]3- ion exhibits a single absorption band at 544 nm. Calculate the crystal field splitting energy ∆ in kJ/mol. In NCS- a stronger or weaker field ligand than water? Predict the color of [Ti(NCS)6]3-.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
0m:0sPlay a video:
85
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 2 videos