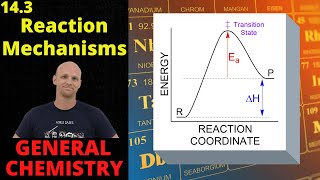
15. Chemical Kinetics
Reaction Mechanism
Multiple Choice
Multiple ChoiceConsider the following elementary steps:
NO2 (g) + NO2 (g) → NO3 (g) + NO (g) [SLOW]
NO (g) + CO3 (g) → N (g) + CO4 (g) [FAST]
What is the rate law of the reaction mechanism?
A
rate = k [NO2]
B
rate = k [NO2]2
C
rate = k [NO][CO3]
D
rate = k [CO3]
E
rate = k [NO4]
1227
views
5
rank
2
comments
Related Videos
Related Practice
Showing 1 of 11 videos