20. Electrochemistry
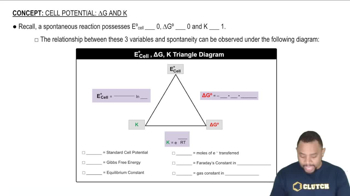
Cell Potential: ∆G and K
Problem 161
Textbook Question
Textbook QuestionExperimental solid-oxide fuel cells that use butane (C4H10) as the fuel have been reported recently. These cells contain composite metal/metal oxide electrodes and a solid metal oxide electrolyte. The cell half-reactions are (b) Use the thermodynamic data in Appendix B to calculate the values of E° and the equilibrium constant K for the cell reaction at 25 °C. Will E° and K increase, decrease, or remain the same on raising the temperature?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
16mPlay a video:
297
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 2 videos