18. Aqueous Equilibrium
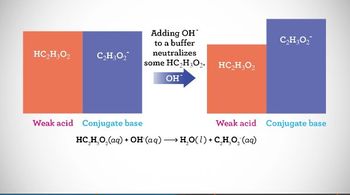
Intro to Buffers
Multiple Choice
Multiple ChoiceA buffer solution is comprised of 50.0 mL of a 0.100 M HC2H3O2 and 60.0 mL of a 0.100 M NaC2H3O2. Which of the following actions would completely destroy the buffer?
A
Adding 0.003 mol HC2H3O2
B
Adding 0.007 mol Ca(C2H3O2)2
C
Adding 0.005 mol NaOH
D
Adding 0.004 mol HCl
E
Adding 0.001 mol HCl
1983
views
3
rank
9
comments
Related Videos
Related Practice
Showing 1 of 16 videos