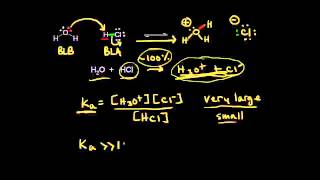
17. Acid and Base Equilibrium
Ka and Kb
Multiple Choice
Multiple ChoiceWhich Bronsted-Lowry acid has the weakest conjugate base?
A
HCNO (Ka = 2.0 x 10-4)
B
HF (Ka = 3.5 x 10-4)
C
HN3 (Ka = 2.5 x 10-5)
D
H2CO3 (Ka = 4.3 x 10-7)
1558
views
4
rank
1
comments
Related Videos
Related Practice
Showing 1 of 8 videos