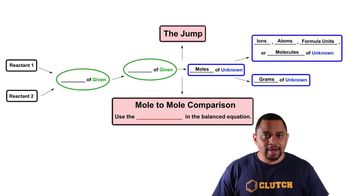
3. Chemical Reactions
Limiting Reagent
Problem 118
Textbook Question
Textbook QuestionThe reaction between ethyl iodide and hydroxide ion in ethanol 1C2H5OH2 solution, C2H5I1alc2 + OH- 1alc2 ¡ C2H5OH1l2 + I - 1alc2, has an activation energy of 86.8 kJ>mol and a frequency factor of 2.10 * 1011 M-1 s-1. (c) Which reagent in the reaction is limiting, assuming the reaction proceeds to completion?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
5mPlay a video:
801
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 11 videos