20. Electrochemistry
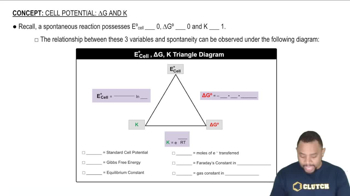
Cell Potential: ∆G and K
Multiple Choice
Multiple ChoiceGiven the reaction: 2 Cl2 (g) + 2 H2O (g) ⇌ 4 HCl (g) + O2 (g) Kp = 7.5x10-2, calculate the Gibbs Free Energy change for the reaction below at 30ºC.
8 HCl (g) + 2 O2 (g) ⇌ 4 Cl2 (g) + 4 H2O (g)
A
-1.3 x 104 J/mol
B
-2.9 x 103 J/mol
C
+4.3 x 105 J/mol
D
+7.7 x 107 J/mol
222
views
Related Videos
Related Practice
Showing 1 of 2 videos