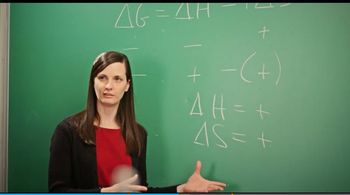19. Chemical Thermodynamics
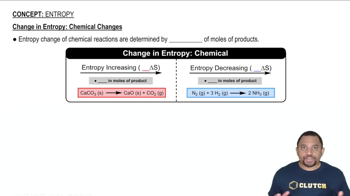
Entropy
Multiple Choice
Multiple ChoiceConsider the freezing of liquid water at 30°C. For this process what are the signs for ∆H, ∆S, and ∆G?
A
∆H = +∆S = -∆G = +
B
∆H = -∆S = +∆G = 0
C
∆H = -∆S = +∆G = -
D
∆H = -∆S = -∆G = +
E
∆H = -∆S = -∆G = -
1818
views
1
rank
4
comments
Related Videos
Related Practice
Showing 1 of 16 videos