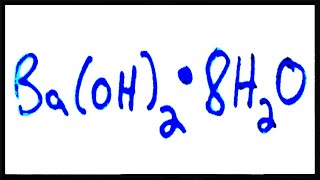3. Chemical Reactions
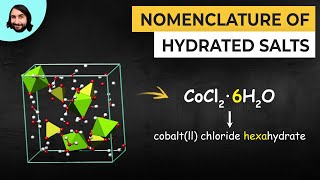
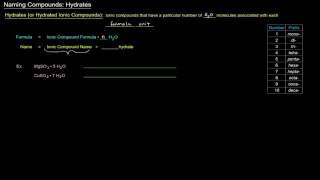
Naming Ionic Hydrates
Problem 134
Textbook Question
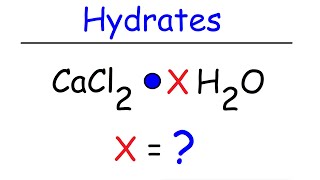
Textbook QuestionA hydrate of copper(II) chloride has the following formula: CuCl2 # x H2O. The water in a 3.41-g sample of the hydrate is driven off by heating. The remaining sample has a mass of 2.69 g. Find the number of waters of hydration (x) in the hydrate.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
0m:0sPlay a video:
3677
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos