9. Quantum Mechanics
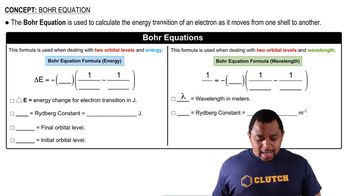
Bohr Equation
Problem 7c2
Textbook Question
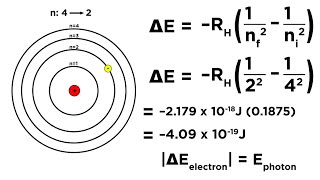
Textbook QuestionConsider the three electronic transitions in a hydrogen atom shown here, labeled A, B, and C. (c) Calculate the wavelength of the photon emitted for each transition. Do any of these transitions lead to the emission of visible light? If so which one(s)?

Calculate the wavelength of the photon emitted for transition B.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
11mPlay a video:
954
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 7 videos