16. Chemical Equilibrium
ICE Charts
Problem 82
Textbook Question
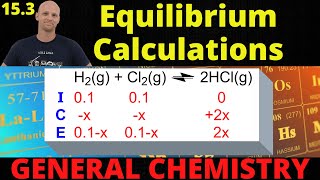
Textbook QuestionA 0.831-g sample of SO3 is placed in a 1.00-L container and heated to 1100 K. The SO3 decomposes to SO2 and O2: 2 SO31g2 Δ 2 SO21g2 + O21g2 At equilibrium, the total pressure in the container is 1.300 atm. Find the values of Kp and Kc for this reaction at 1100 K.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
13mPlay a video:
1218
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 12 videos