9. Quantum Mechanics
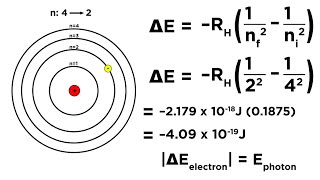
Bohr Model
Multiple Choice
Multiple ChoiceWhich of the following statements about the Bohr model of the hydrogen atom is false?
A
The energy of each Bohr orbit is quantized.
B
An electron in a stationary state does not emit radiation.
C
Transitions between stationary states that are close together require shorter wavelength that those that are spaced further apart.
D
When an electron jumps to a higher state, energy is absorbed.
E
When an electron falls to a lower state, energy is emitted.
F
All of the above are correct.
305
views
Related Videos
Related Practice
Showing 1 of 10 videos