9. Quantum Mechanics
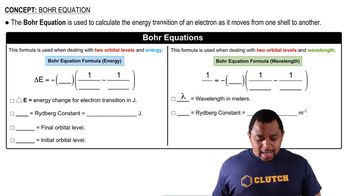
Bohr Equation
Problem 64
Textbook Question
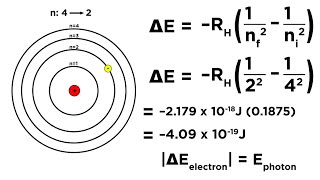
Textbook QuestionUse the Balmer equation to calculate the wavelength in nano-meters of the spectral line for hydrogen when n = 6 and m = 2. What is the energy in kilojoules per mole of the radiation corresponding to this line?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
1048
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 7 videos