18. Aqueous Equilibrium
Intro to Buffers
Problem 4a
Textbook Question
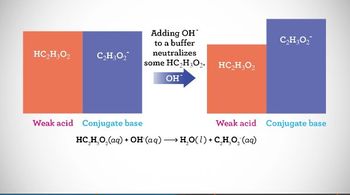
Textbook QuestionThe following diagram represents a buffer composed of equal concentrations of a weak acid, HA, and its conjugate base, A-. The heights of the columns are proportional to the concentrations of the components of the buffer. (c) Which of the three represents a situation that cannot arise from the addition of either an acid or a base? [Section 17.2]

 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
686
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 16 videos