7. Gases
Partial Pressure
Multiple Choice
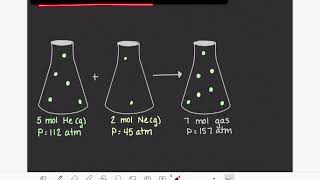
Multiple ChoiceA 2.50-L container contains a mixture of helium, nitrogen, and argon gas. The total pressure is 1.800 atm at 295 K. The partial pressure of helium is 0.650 atm, and the partial pressure of nitrogen is 0.350 atm. How many moles of argon gas are present in the mixture?
A
0.800 moles
B
0.074 moles
C
0.083 moles
D
0.186 moles
E
0.444 moles
329
views
Related Videos
Related Practice
Showing 1 of 13 videos