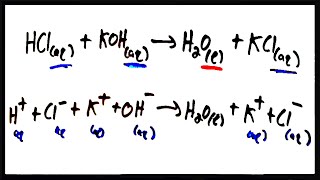6. Chemical Quantities & Aqueous Reactions
Complete Ionic Equations
Multiple Choice
Multiple ChoiceWhen 100.0 mL of a solution that is 0.00100 M in Cu(NO3)2 is mixed with 100 mL of a 0.20 M solution of NH3, what concentration of When 100.0 mL of a solution that is 0.00100 M in Cu(NO3)2 is mixed with 100 mL of a 0.20 M solution of NH3, what concentration of Cu2+ remains in solution? remains in solution?
A
5.0 × 10−4M
B
2.9 × 10−13M
C
3.7 × 10−14M
D
2.9 × 10−16M
255
views
Related Videos
Related Practice
Showing 1 of 11 videos