18. Aqueous Equilibrium
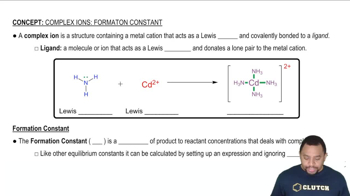
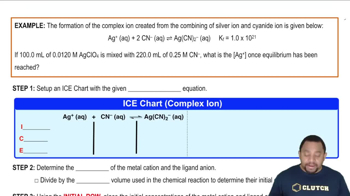
Complex Ions: Formation Constant
Problem 120
Textbook Question
Textbook QuestionDissolution of 5.0 x 10^-3 mol of Cr(OH)3 in 1.0 L of 1.0 M NaOH gives a solution of the complex ion [Cr(OH)4]- (Kf = 8 x10^29). What fraction of the chromium in such a solution is present as uncomplexed Cr3+?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
12mPlay a video:
517
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 2 videos