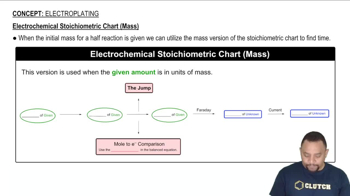
20. Electrochemistry
Electroplating
Multiple Choice
Multiple ChoiceCu2+ is reduced to Cu(s) at an electrode. If a current of 1.25 A is passed for 72 hours, what mass of copper is deposited at the electrode? (MW of Cu: 63.55 g/mol)
A
91.5 g
B
55.8 g
C
83.1 g
D
110 g
188
views
1
comments
Related Videos
Related Practice
Showing 1 of 6 videos