20. Electrochemistry
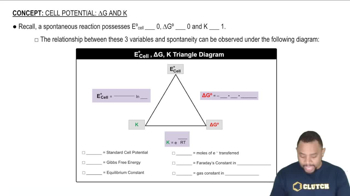
Cell Potential: ∆G and K
Problem 122a
Textbook Question
Textbook QuestionCalculate ∆Gr°xn and K for each reaction. b. The reaction of Cr3+(aq) and Cr(s) to form Cr2+(aq). [The electrode potential of Cr2+(aq) to Cr(s) is -0.91 V.]
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
12mPlay a video:
774
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 2 videos