12. Molecular Shapes & Valence Bond Theory
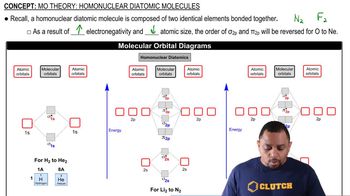
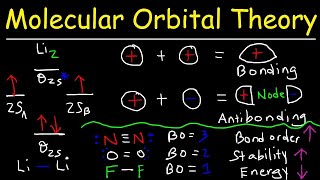
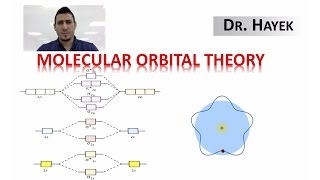
MO Theory: Homonuclear Diatomic Molecules
Multiple Choice
Multiple ChoiceWhich of the following statements is true about polarity?
A
All molecules with polar bonds are polar.
B
A nonpolar molecule has only nonpolar bonds.
C
The shape of a molecule is important in determining the molecular polarity.
D
The greater the difference in electronegativity between two atoms in a bond, the smaller the magnitude of polarity.
E
All of the above are true statements.
621
views
Related Videos
Related Practice
Showing 1 of 8 videos