13. Liquids, Solids & Intermolecular Forces
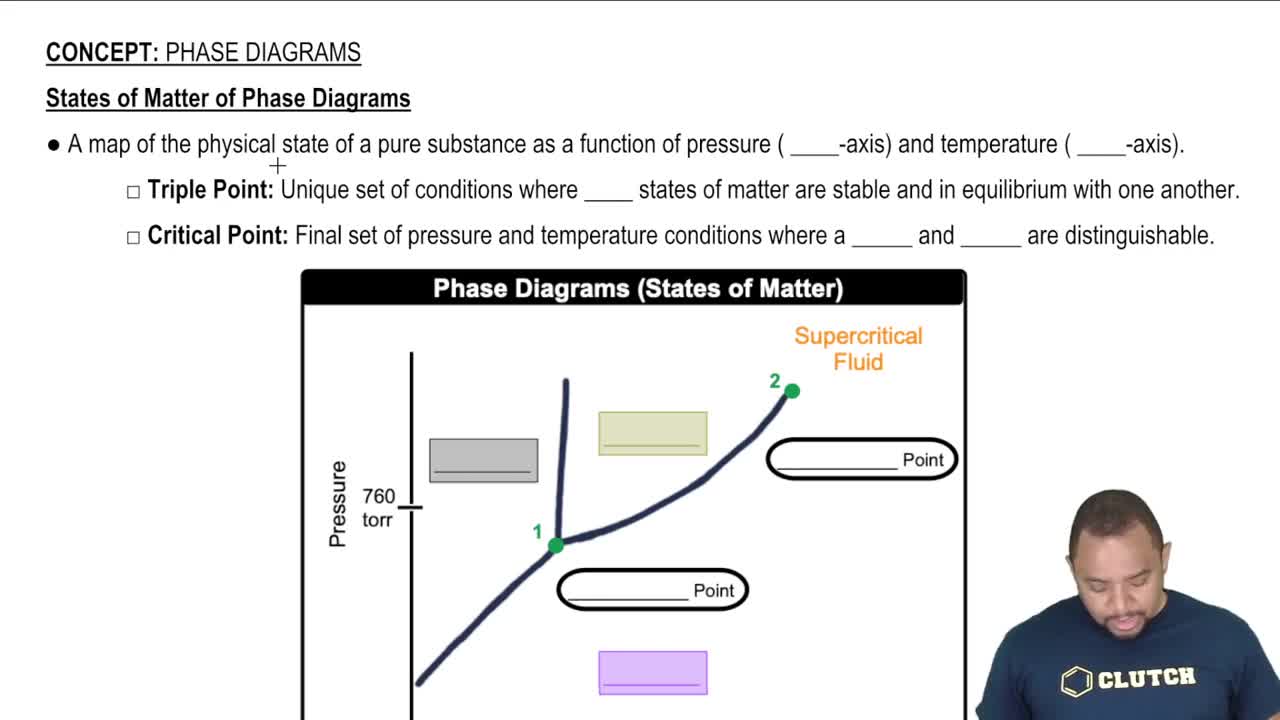
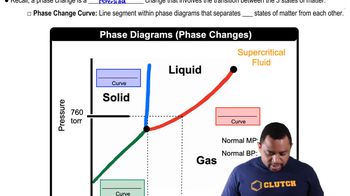
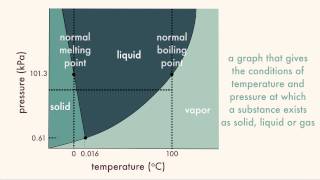
Phase Diagrams
Multiple Choice
Multiple ChoiceCalculate the amount of heat (in J) required to heat 2.00 grams of ice from
−12.0℃ to 88.0℃.
The heat of fusion of water is 6.02 kJ/mol, the specific heat capacity of ice is 2.09 J/g ▪℃ and the specific heat capacity of water is 4.184 J/g ▪℃.
A
50.2 J
B
418 J
C
668 J
D
736 J
E
1454 J
301
views
Related Videos
Related Practice
Showing 1 of 12 videos