18. Aqueous Equilibrium
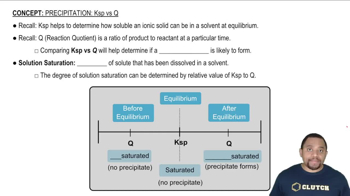
Precipitation: Ksp vs Q
Problem 46
Textbook Question
Textbook QuestionThe following pictures represent solutions of Ag2CrO4, which also may contain ions other than Ag+ and CrO42- that are not shown. Solution 1 is in equilibrium with solid Ag2CrO4. Will a precipitate of solid Ag2CrO4 form in solutions 2-4? Explain.

 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
581
views
1
rank
Was this helpful?
Related Videos
Related Practice
Showing 1 of 2 videos