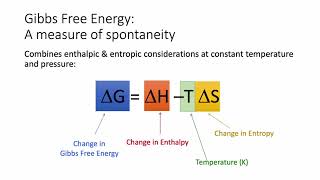
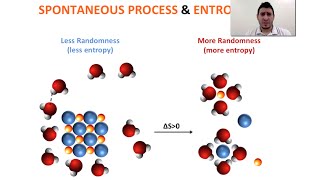
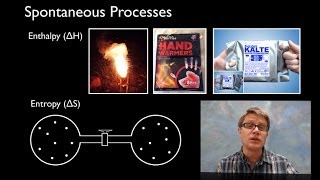
19. Chemical Thermodynamics
Spontaneous vs Nonspontaneous Reactions
Multiple Choice
Multiple ChoiceWhich of the following is a true statement about ΔSsurroundings?
A
ΔSsurroundings is positive when the system absorbs heat from the surroundings.
B
ΔSsurroundings will decrease as a result of an exothermic process.
C
ΔSsurroundings is always positive when ΔSuniv is positive.
D
ΔSsurroundings is independent of the heat released or absorbed by the system.
E
ΔSsurroundings is always positive when ΔSsystem is negative for a spontaneous process.
220
views
Related Videos
Related Practice
Showing 1 of 9 videos