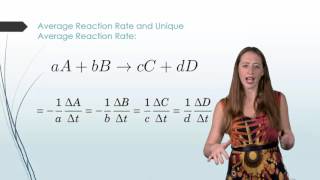15. Chemical Kinetics
Average Rate of Reaction
Problem 19b
Textbook Question
Textbook QuestionConsider the following hypothetical aqueous reaction: A1aq2S B1aq2. A flask is charged with 0.065 mol of A in a total volume of 100.0 mL. The following data are collected: Time (min) 0 10 20 30 40 Moles of A 0.065 0.051 0.042 0.036 0.031 (b) Calculate the average rate of disappearance of A for each 10-min interval in units of M>s.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
11mPlay a video:
1546
views
1
rank
Was this helpful?
Related Videos
Related Practice
Showing 1 of 8 videos