1. Intro to General Chemistry
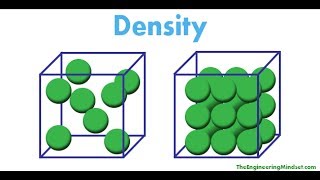
Density
Problem 35b
Textbook Question
Textbook Question(b) Carbon dioxide 1CO22 is a gas at room temperature and pressure. However, carbon dioxide can be put under pressure to become a 'supercritical fluid' that is a much safer dry-cleaning agent than tetrachloroethylene. At a certain pressure, the density of supercritical CO2 is 0.469 g>cm3. What is the mass of a 25.0-mL sample of supercritical CO2 at this pressure?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
1647
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 9 videos