11. Bonding & Molecular Structure
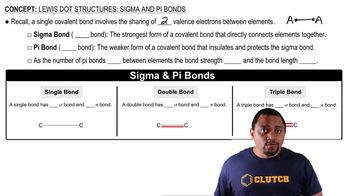
Lewis Dot Structures: Sigma & Pi Bonds
Problem 102
Textbook Question
Textbook QuestionButadiene, C4H6, is a planar molecule that has the following carbon–carbon bond lengths:

 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
862
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 7 videos