19. Chemical Thermodynamics
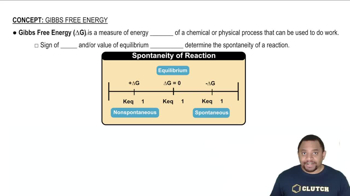
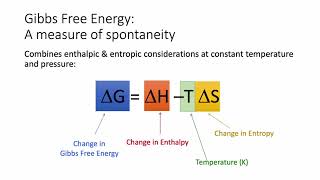
Gibbs Free Energy
Problem 63b
Textbook Question
Textbook QuestionUse standard free energies of formation to calculate ΔG° at 25 °C for each reaction in Problem 61. How do the values of ΔG° calculated this way compare to those calculated from ΔH° and ΔS°? Which of the two methods could be used to determine how ΔG° changes with temperature?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
0m:0sPlay a video:
2634
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos