19. Chemical Thermodynamics
Entropy
Problem 88
Textbook Question
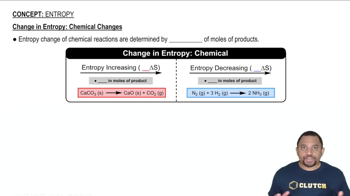
Textbook QuestionFor each of the following processes, indicate whether the signs of ΔS and ΔH are expected to be positive, negative, or about zero. (e) A piece of charcoal is combusted to form CO21g2 and H2O1g2.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
776
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 16 videos