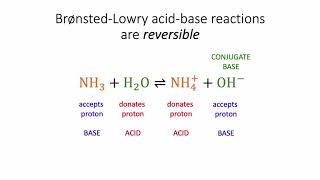17. Acid and Base Equilibrium
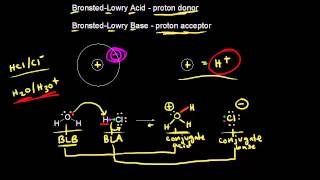
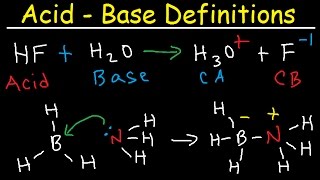
Bronsted-Lowry Acids and Bases
Problem 21e
Textbook Question
Textbook QuestionLabel each of the following as being a strong base, a weak base, or a species with negligible basicity. In each case write the formula of its conjugate acid, and indicate whether the conjugate acid is a strong acid, a weak acid, or a species with negligible acidity: (a) CH3COO-
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
657
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 15 videos