18. Aqueous Equilibrium
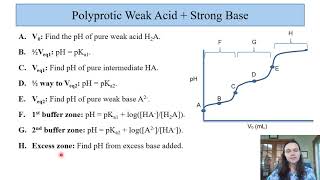
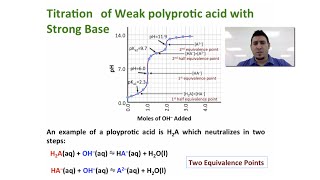
Titrations: Diprotic & Polyprotic Buffers
Problem 147
Textbook Question
Textbook QuestionEthylenediamine (NH2CH2CH2NH2, abbreviated en) is an organic base that can accept two protons: (a) Consider the titration of 30.0 mL of 0.100 M ethylenediamine with 0.100 M HCl. Calculate the pH after the addition of the following volumes of acid, and construct a qualitative plot of pH versus milliliters of HCl added: (i) 0.0 mL (ii) 15.0 mL (iii) 30.0 mL (iv) 45.0 mL (v) 60.0 mL (vi) 75.0 mL
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
23mPlay a video:
1124
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos