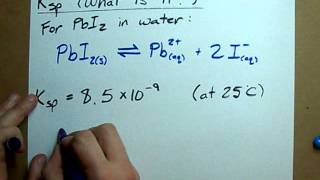18. Aqueous Equilibrium
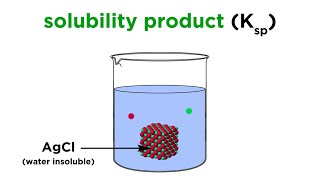
Solubility Product Constant: Ksp
Problem 100
Textbook Question
Textbook QuestionTooth enamel is composed of hydroxyapatite, whose simplest formula is Ca51PO423OH, and whose corresponding Ksp = 6.8 * 10-27. As discussed in the Chemistry and Life box on page 746, fluoride in fluorinated water or in toothpaste reacts with hydroxyapatite to form fluoroapatite, Ca51PO423F, whose Ksp = 1.0 * 10-60. (a) Write the expression for the solubility-constant for hydroxyapatite and for fluoroapatite.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
4mPlay a video:
1658
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos