11. Bonding & Molecular Structure
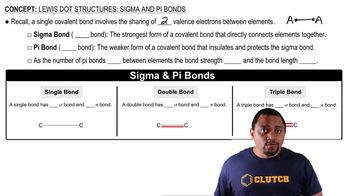
Lewis Dot Structures: Sigma & Pi Bonds
Problem 7a
Textbook Question
Textbook QuestionThe partial Lewis structure that follows is for a hydrocarbon molecule. In the full Lewis structure, each carbon atom satisfies the octet rule, and there are no unshared electron pairs in the molecule. The carbon—carbon bonds are labeled 1, 2, and 3. (c) Which carbon—carbon bond is the strongest one?

 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
338
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 7 videos