In this video, we're going to talk about how to calculate the isoelectric point of a peptide. So, recall from our previous lesson videos on the isoelectric point that the isoelectric point, or the PI calculation, is always just going to be the midpoint or the average between the 2 pKa values for the 2 ionizations involving the neutral species. And so, all this is saying is that, whenever we're calculating the isoelectric point, we're only going to average 2 pKa values, regardless of how many pKa values were given, and the same is going to apply for calculating the isoelectric point of a peptide. And so, before we get to our example below, there's really 2 ideas that I really want to emphasize. And the first is a similar idea to the one that we talked about when we were calculating the net charge of a peptide. And this is the idea that the isoelectric point and the net charge of a peptide can both only be estimated. They can only be estimated, and this is because of the unique microenvironment that each residue has in a peptide. And so, the true PI values can only be experimentally determined. And because we know that the microenvironment is capable of shifting the pKa values of residues, you need to make sure that you're using the correct set of pKa values, specifically the set of pKa values for amino acid residues and not for pKa values of free amino acids. And so if your professor uses the same set of pKa values for amino acid residues and free amino acids, then you're good to go and you don't have to worry about this. But just in case, you should double-check to make sure that your professor doesn't want you to use a specific set of pKa values for amino acid residues. And so when we're doing our practice problems, we're going to be using a specific set of pKa values for amino acid residues. And so the second idea that I really want to emphasize is just this idea that the isoelectric point of a peptide follows similar steps for calculating the isoelectric point of amino acids with ionizable R groups. And so this little, chart here, this little diagram might look familiar to you because it's really the same one from our previous lesson. And so let's take a look at the 4 steps that we need to follow to calculate the isoelectric point of a peptide. And again, the first step is just going to be to know how the amino acid R groups ionize. And really all this is saying is that we need to know the 7 amino acids with ionizable R groups. And recall that our mnemonic to help us remember those 7 amino acids is just "yucky crazy dragons knights riding horses". And so if you know this mnemonic and how it works, then you'll know those 7 amino acid, with ionizable R groups and you'll be set to go with step number 1. Now step number 2 is going to be to order the pKa values from the smallest to the greatest. And so here, we're showing only 4 pKa values, but regardless of how many pKa values there are, there could be 5, there could be 6, there could be 8, however many there are, you're gonna want to, order them all from the smallest to the largest. And so step number 3 is going to be to determine the net charge of predominant structures at any pH between each pair of adjacent pKa values or neighboring pKa values. And so what you can see down here is that we've got our 4 pKa values ordered from smallest to greatest and then, we have these, pH ranges in between the pKa values that we're going to check for the predominant structure. And we can either imagine the predominant structure, so we don't need to draw it out unless it tells us to. Or if you're still working on, if you're having trouble imagining it, you can always draw the predominant structures. But the whole point is just to determine the net charges of the predominant structures, and so we've got these ranges in between each pKa that we need to check. And so, we would check each one of these. And so the way that you determine the net charge, again, of the predominant structure, is by comparing the pH of the solution that we choose to the pKa values of all the ionizable groups. And of course, the last step is going to be to calculate the isoelectric point just by averaging the 2 pKa values, so it's always just going to be 2. It's gonna be the 2 pKa values that sandwich the pH where the predominant structure has a neutral net charge. So again, the isoelectric point is going to be the pH where the structure is going to have a neutral net charge or a net charge of 0. And so whatever 2 pKa values they are, so if it were this one and this one, you would average these 2 pKa values if this structure here had a neutral net charge. If it wasn't these two pKa values, if it was this pK and this pKa because this structure had a neutral net charge, then you would average these 2 pKa values here. So it's all about where do you find that neutral net charge. And so, let's go ahead and move on to our example. And in this example, it says to estimate the isoelectric point for the following peptide, DGE, which is aspartic acid, glycine, and glutamic acid. And so, all we're gonna do here is consider all of the ionizable groups. So we don't really care about any of the other groups that are not ionizable because they're not gonna contribute to the net charge, and so if they don't contribute to the net charge, then it's not gonna contribute to the isoelectric point. And so for D, G, and E, we can rewrite it over here. And so we basically got a few ionizable groups that we need to consider. We know that the first amino acid residue in a chain is gonna have an ionizable amino group, so there's an ionizable amino group that we need to consider. Now, aspartic acid is going to be one of our negatively charged acetic amino acid R groups. So we know that its r group potentially could have a negative charge, so we'll also leave it as a blank here to consider its charge. Now, glycine does not have an ionizable R group. It's not one of our 7 amino acids with ionizable R-groups, so we don't care about its R group and it doesn't have any ionizable amino or carboxyl groups because it's an internal amino acid residue. Now, glutamic acid over here does have an ionizable R group that's potentially negative, so we are going to consider that. And then also because it's the last residue in the chain, it also has a potentially negative carboxyl group. And so all we need to do is, now that we've done step number 1, we know how these amino acid R groups ionize and we've even highlighted them like this to consider their ionizations. Step number 2 is gonna be to order the pKa values for all 4 of these ionizable groups in order from smallest to largest. So now, we're going to use our pKa set over here for amino acid residues, and we'll start with the amino group over here, and the amino group is gonna be the N-terminus. And so, notice that the N-terminus has a pKa of 8, and the pKa of 8 is, when we compare it to all of the other pKa values that we're going to be looking at, we have glutamic acid, for glutamic acid, has a pKa of 4.1. We have aspartic acid, which is the D here, has a pKa of 3.9. And then we also have the carboxyl group, and the carboxyl group is the C-terminus up here, which has a pKa of 3.5. So we have our 4 pKa values, we need to order them from smallest to largest, and so the smallest one is the pKa of the C-terminus. So we'll put 3.5 over here, and we'll put carboxyl above it to remind us it's the carboxyl group. The next one is aspartic acid, so aspartic acid is 3.9 and this is we'll put ASP for aspartic acid to R group. The next one is going to be glutamic acid, which is 4.1. So we'll put 4.1 in a Glu for glutamic acid. And then, of course, our last one's going to be the N-terminus, which is 8.0, and so this is for the amino group. And so, again, all we need to do is choose any range. We can choose this range here or this range here or this range here, it doesn't matter. You can start with anyone that you want. Let's start with the one on the left since we normally do things from left to right anyway. So let's choose this range right here. We can pick any pH that we want as long as it falls into this range, as long as it falls between these 2 pKa values. So let's just choose, I don't know, 3.6, pH of 3.6. And so all we need to do is imagine the predominant structure at pH 3.6 and determine the net charge of the predominant structure at pH 3.6. And so all we need to do is compare the pH to each of the pKa values. So let's start with the amino group. So with the amino group, the pKa is 8. And so a pH of 3.6 is less than the pKa of 8. And when the pKa is greater than the pH, that means that the conjugate acid's going to predominate. And the conjugate acid of the amino group is an NH3+ with a positive charge. So up here, what we're gonna do for our amino group is we're gonna put a plus for the positive charge to consider the charge. So now, let's move on to aspartic acid since that's next in our chain here. Aspartic acid's R group. And so we compare the pH to aspartic acid's R group which is 3.9. And a pH of 3.6 is less than the pH of 3.9, so that means that the conjugate acid form of aspartic acid's R group is going to predominate. And aspartic acid's conjugate acid actually has a neutral charge. So it has a charge of 0. So in here, what we can do is just put a 0 because it has a neutral net charge at pH 3.6. So then we can check, glutamic acid's R group, which is here since it's next in our chain, and glutamic acid's R group is here, its pKa is 4.1. And so, a pH of 3.6 is less than the pH of 4.1. When the pH is less than the pKa, the conjugate acid predominates. The conjugate acid of glutamic acid has a neutral net charge of 0. So we'll just put a 0 in here. And then, we take a look at the carboxyl group, which is last and the carboxyl group, as you can see, which is, way over here, it's pKa is 3.5. And the pKa of 3.5 is less than the pH of 3.6. And when the pH is greater than the pKa, the conjugate base predominates and the conjugate base of the carboxyl group is going to be a carboxylate anion, oops, with a negative charge. We'll put this in blue. And so, all we need to do is total the net charges of each of these ionizable groups to get the net charge of, the structure at pH 3.6. So we have a positive and a negative. So this group over here cancels out with this group over here, and then the other two groups are neutral. So that means that at this pH range, at any pH in here, our structure has a neutral net charge of 0. And so recall that, really when we're calculating the PI, we want to find the, pH range that has a neutral net charge of 0. So we were able to find it on our first try. Now, if we were to choose a different range, if we had selected a different range GCBOVER HERE REMEMBER THAT, WHEN YOU CROSS A pKa, that the net charge of the predominant structure changes by just one unit. And when you go from left to right, it goes down one charge. And so from going from this yellow range over to this green range, and crossing this pKa here, we know that the predominant structure of the net charge is going to change. So any pH in this range, it's going to go down by 1, so we know it would be negative one over here. And so if we had chosen this pH range, then all we would have done is done the same cheat sheet, but we would have gone the opposite direction over here because we know that the charge would go up by 1, and that would give us a net charge of 0. So it doesn't really matter which pH range that you choose. You choose 1, determine the net charge, and then you can use our cheat sheet here to determine the net charge in a different range. So now that we found our neutral net charge, we have to average the 2 pKa values sandwiching the neutral net charge. So the 2 pKa values sandwiching it are this one here and this one here. So we can do our PI calculation over here, and it's just going to be the average of these two numbers. So 3.9 plus 3.5 divided by 2. And so this comes out to 7.4 divided by 2, which comes out to 3.7. And what you'll see is that the pI of 3.7 matches our answer option A. So that means that A here is correct, we can give it a check, indicate that A here is correct, and all our other ones are incorrect, so we can cross them off. So this concludes our example here in our lesson and we'll get some practice calculating the isoelectric point of peptides in our practice. So I'll see you guys in those videos.
4. Protein Structure
Isoelectric Point of a Peptide
4. Protein Structure
Isoelectric Point of a Peptide - Online Tutor, Practice Problems & Exam Prep
1
concept
Isoelectric Point of a Peptide
Video duration:
13mPlay a video:
Video transcript
2
Problem
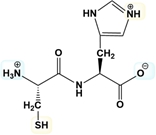
ProblemEstimate the isoelectric point of the following dipeptide in the figure.
A
4.75
B
7
C
9.5
D
10.5
3
Problem
ProblemCalculate the approximate pI of the peptide: C-G-E-K.
A
3.08
B
5.71
C
6.05
D
9.63
4
Problem
ProblemDraw in the R-groups for the following peptide & calculate the pI: ATLDAG.
A
7.41
B
6.9
C
9.25
D
3.7