Alright. In this video, we're going to explore our first category of lipids, the fatty acids. And so first notice that down below over here, we have a different version of our original lipid map. And you can see that the lipids are going to be broken up into the fatty acid based lipids, which is this entire left branch over here and then we also have the isoprene based lipids and notice that the Isoprene based lipids are being abbreviated with this Isoprene molecule and we're going to continue to do that until after we've explored all of the fatty acid lipids in their entirety. And then again, we'll go and explore the isoprenes last. And so what in the world are these fatty acids? Well, fatty acids, which are also sometimes generally called just fats occasionally, these are just hydrocarbon chains of varying length that have a terminal carboxylic acid group. And so if we take a look at our image down below right here, notice we have a fatty acid molecule. We have this long hydrocarbon chain, and at the very end of the hydrocarbon chain, we have this carboxylic acid, just as we described up above. Now, these fatty acids are gonna be used as building blocks for more complex lipids such as glycerolipids, sphingolipids, and all of these other ones down below, waxes and eicosanoids as well. And so, what's important to note is that the carbon atoms of fatty acids, they're usually numbered, starting at the carboxyl carbon atom. And so notice over here we have the carboxyl carbon atom, and so this would be carbon number 1. And what's also important to note is that the alpha carbon designated by just the Greek symbol alpha is going to be the carbon atom that is adjacent or right next to the carboxyl carbon atom. And so this carbon atom right here in blue, because it's adjacent or right next to the carboxyl carbon atom, this is going to be the alpha carbon atom, and you can think the a in alpha is just for the a in adjacent right next to. And then, of course, the omega carbon atom symbolized by the Greek letter omega is going to be the carbon atom that is furthest from the carboxyl carbon atom. And so, of course, this carbon atom over here is the one that's furthest away from the carboxylic carbon. And so this is going to be the Omega Carbon. And so what's important to note is that before we actually move on to talking about Glycerolipids, we're we're actually gonna talk about fatty acids for a little while. We're gonna talk about different types of fatty acids such as unsaturated and saturated in our next lesson video. And then we'll talk about nomenclature and some other fatty acids as well. So we're gonna stick on this fatty acids for a little bit before we actually get to glycerolipids. And so this here concludes our introduction to fatty acids and again, in our next lesson video we'll be able to talk about unsaturated and saturated fatty acids. So I'll see you guys there.
Fatty Acids - Online Tutor, Practice Problems & Exam Prep
Fatty Acids
Video transcript
Which fatty acid chain would you expect to be least soluble in water?
a) 16-Carbon fatty acid.
b) 4-Carbon fatty acid.
c) 22-Carbon fatty acid.
d) 12-Carbon fatty acid.
Fatty Acids
Video transcript
In this video, we're going to differentiate between saturated and unsaturated fatty acids. These fatty acids that we've been discussing can be categorized into 2 different groups based on the nature of their hydrocarbon chains. These two groups are the saturated fatty acids and the unsaturated fatty acids. Now, as the name implies, the saturated fatty acids are going to be fully saturated with hydrogen atoms. That means their hydrocarbon chains will only contain Carbon-Carbon single bonds, and there will not be any Carbon-Carbon double bonds. If we take a look at our image down below, over here on the left-hand side, notice we're showing you an example of a saturated fatty acid. You can see that it's got a carboxylic acid group and this long hydrocarbon chain, which only has Carbon-Carbon single bonds, meaning the hydrocarbon chain is fully saturated with hydrogen. Over here, we have a different 3D representation of the same exact fatty acid, and this is actually the fatty acid stearate, which is one of the most abundant saturated fatty acids in animals.
Now, the second group of fatty acids are the unsaturated fatty acids. As their name implies, they are not going to be fully saturated with hydrogen atoms. This is due to the presence of greater than or equal to 1 Carbon-Carbon double bond in the hydrocarbon chain. The prefixes mono- and poly- respectively indicate the presence of just 1 or more than 1 double bond. The double bonds present in unsaturated fatty acids are almost always in the cis conformation, which creates kinks in the chains of unsaturated fatty acids. If we take a look at our image down below, over here, notice we're showing another fatty acid, but this one is an unsaturated fatty acid because there's the presence of a double bond. Notice that the double bond is almost always in the cis conformation, which creates a kink in the chain of the unsaturated fatty acid. Because there's only one double bond present here in this specific unsaturated fatty acid, oleate, this makes it a monounsaturated fatty acid, indicating the presence of just one double bond. Over here on the right, we have another unsaturated fatty acid, linoleate, which has the presence of 3 double bonds. Because it has more than one double bond, this would be a polyunsaturated fatty acid. The double bonds are almost always in the cis conformation. This creates kinks in the chains of unsaturated fatty acids, and that will be important in our next lesson video when we talk about the melting points of these fatty acids.
This here concludes our introduction to saturated and unsaturated fatty acids, and we'll be able to get some practice applying these concepts in our next video. So I'll see you guys there.
What is the molecular formula of Linolenic acid, an 18-carbon polyunsaturated fatty acid with 3 double bonds?
a) C18H32O2
b) C18H30O2
c) C28H30O2
d)C18H34O2
Fatty Acids
Video transcript
In this video, we're going to talk about the melting points of fatty acids, and really there are two primary factors that affect the strength of the hydrophobic interactions between the hydrocarbon chains of fatty acids, and the strength of the hydrophobic interactions is actually what affects the melting point of the fatty acids. And so these two factors, by affecting the strength of the hydrophobic interactions, they're also affecting the melting point of the fatty acids. And so the first of these two primary factors is going to be the length of the hydrocarbon chains or the amount of carbon atoms that are present in the hydrocarbon chains. And so the longer these hydrocarbon chains are, the higher the melting point will actually be. And so if we take a look down below at our image at table number 1, which corresponds with number 1 up above, you'll notice that we have the fatty acid name in the first column and the second column practically has the number of carbon atoms or the length of the hydrocarbon chain and the 3rd column has the corresponding melting point in degrees Celsius. And you'll notice that as we increase the number of carbon atoms in the hydrocarbon chain or as we increase the length of the hydrocarbon chain, the melting point is also increasing and so, it's true that the longer the chain is, the higher the melting point will be.
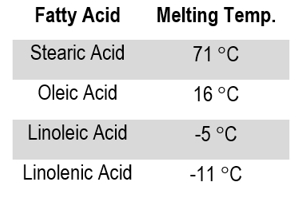
Now, the second primary factor that affects the strength of hydrophobic interactions, and thus the melting point of the fatty acids, is going to be the degree of saturation of the hydrocarbon chains or the amount of double bonds that are present in the hydrocarbon chain. And so this time, it turns out that the more double bonds there are in the hydrocarbon chain, the lower the melting point is going to be. And so if we take a look at our second table, table number 2, which corresponds with number 2 up above. Notice again, we have the fatty acid chain name in the first column and then we have the degree of saturation or the number of double bonds in the second column and again, the melting point in degrees Celsius in the 3rd column. And notice that this time, as we increase the number of double bonds, that the melting point is actually decreasing. And so we start off with positive 16 degrees, and we go down to negative 50 degrees. So, the melting point temperature is actually decreasing as we increase the number of double bonds. Just like what we said up above. The more double bonds, the lower the melting point. And so, therefore, because this here is true. This means that unsaturated fatty acids, which are gonna end up having kinks in their chain due to the double bonds, are gonna also end up having lower melting points than saturated fatty acids.
And so if we take a look at our image over here, what you'll note is on the left hand side, we have saturated fatty acids and on the right hand side, what we have are unsaturated fatty acids. And so taking a look at the, saturated fatty acids over here on the left hand side, notice that they are pretty highly packed. And that's because they don't have any double bonds and they don't have any kinks in their chain. And so their fatty acid chains are able to be pretty linear and, they're able to be pretty highly packed. And they're so highly packed, the more packing there is, the more hydrophobic interactions that can form between all of these hydrophobic chains. Higher the higher the melting point will be. Now, on the other hand, taking a look at the unsaturated fatty acids, which again are gonna have these double bonds, the double bonds are ultimately going to create kinks in the chains of these unsaturated fatty acids. And so notice that these chains are not linear, they're actually, forming these kinks and that makes them a lot harder to pack tightly so they are going to be less packed and if they're less packed, that means that they're going to be less hydrophobic interactions forming between them and less hydrophobic interactions corresponds with having a lower melting point.
Now, a higher melting point means that at room temperature, these are are going to be solids at room temperature such as butter, which are again solids at room temperature. And on the other hand, having a lower melting point means that you're gonna melt a lot easier at room temperature and so they're going to be liquids at room temperature such as oils. And so down below, we've defined fats as a general term referring to lipids or fatty acids that are solids at room temperature and then, of course, oils are going to be liquids at room temperature. And what you'll notice is that the, fats such as butter here, they're typically extracted from animals and that's because although animals have both saturated and unsaturated fatty acids, they tend to have a higher percentage of saturated fatty acids. And so fats are typically extracted from animals. And on the other hand, plants, and oils, I'm sorry, oils which are liquids at room temperature, they tend to be extracted from plants and that's because plants, again, although they have both saturated and unsaturated fatty acids, they tend to have a higher percentage of unsaturated fatty acids, and that's what allows, oils to be extracted from plants, typically. And so this here concludes our lesson on the melting points of fatty acids and we'll be able to get some practice applying all of these concepts in our next couple of practice problems. So I'll see you guys there.
What aspect of each of the 18-carbon fatty acids in the table below is correlated with their melting point?
a) The charge of the carboxylic acid group.
b) The length of the hydrocarbon chain
c) The number of double bonds
d)The polar hydrocarbon chains
What happens to the melting point in fatty acids as the hydrocarbon length increases?
a) It increases.
b) It decreases.
c) It stays the same.
d) There is no direct correlation.
Unsaturated fatty acids:
a) Usually contain a double bond with cis stereochemistry.
b) Are found in both plants and animals.
c) Sometimes contain multiple double bonds.
d) Have lower melting points than the analogous saturated fatty acids.
e) All of the above are correct.
Which of the following are correct with regard to saturated fatty acids?
a) They are generally solid at room temperature.
b) The carbon backbone contains at least one double bond, creating a kink in the chain.
c) Come primarily from vegetable products.
d) They are only hydrocarbon chains, making them liquids at room temperature.
e) Cannot be present in other lipids, such as phospholipids.
Match each of the fatty acids with the appropriate melting point:

a) CH3(CH2)18COOH ______
b) CH3(CH2)14COOH ______
c) CH3(CH2)10COOH ______
d) CH3(CH2)7CH=CH(CH2)7COOH ______
e) CH3(CH2)4CH=CH-CH2-CH=CH(CH2)7COOH ______