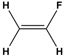
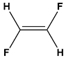
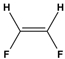
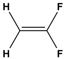
We're going to quickly cover chemical bonds. So you guys already know that chemical bonds are how individual atoms interact with one another and how they link and connect to other atoms. And really, chemical bonds fall under one of two categories, either intramolecular bonds or intermolecular bonds. Now, intramolecular bonds are bonds that exist within a molecule, whereas intermolecular bonds exist between different molecules. And so these two words sound similar, and what helps me distinguish between them is that intramolecular bonds are trapped within a molecule, whereas intermolecular bonds are not trapped, and instead, they exist between different molecules. So, in our example below, we're going to label the chemical bonds appropriately. And notice we have two different HCl molecules down below, and this bond here between the H and the Cl, the chlorine atoms, this is trapped within the molecule. So this must be our intramolecular bond. Now, the other bond shown here with the dotted line that exists between two different HCl molecules is not trapped, so this is an intermolecular bond. In our next video, we're going to talk about specific intramolecular bonds, and that includes ionic and covalent bonds. So I'll see you guys in that video.
- 1. Introduction to Biochemistry4h 34m
- What is Biochemistry?5m
- Characteristics of Life12m
- Abiogenesis13m
- Nucleic Acids16m
- Proteins12m
- Carbohydrates8m
- Lipids10m
- Taxonomy10m
- Cell Organelles12m
- Endosymbiotic Theory11m
- Central Dogma22m
- Functional Groups15m
- Chemical Bonds13m
- Organic Chemistry31m
- Entropy17m
- Second Law of Thermodynamics11m
- Equilibrium Constant10m
- Gibbs Free Energy37m
- 2. Water3h 23m
- 3. Amino Acids8h 10m
- Amino Acid Groups8m
- Amino Acid Three Letter Code13m
- Amino Acid One Letter Code37m
- Amino Acid Configuration20m
- Essential Amino Acids14m
- Nonpolar Amino Acids21m
- Aromatic Amino Acids14m
- Polar Amino Acids16m
- Charged Amino Acids40m
- How to Memorize Amino Acids1h 7m
- Zwitterion33m
- Non-Ionizable Vs. Ionizable R-Groups11m
- Isoelectric Point10m
- Isoelectric Point of Amino Acids with Ionizable R-Groups51m
- Titrations of Amino Acids with Non-Ionizable R-Groups44m
- Titrations of Amino Acids with Ionizable R-Groups38m
- Amino Acids and Henderson-Hasselbalch44m
- 4. Protein Structure10h 4m
- Peptide Bond18m
- Primary Structure of Protein31m
- Altering Primary Protein Structure15m
- Drawing a Peptide44m
- Determining Net Charge of a Peptide42m
- Isoelectric Point of a Peptide37m
- Approximating Protein Mass7m
- Peptide Group22m
- Ramachandran Plot26m
- Atypical Ramachandran Plots12m
- Alpha Helix15m
- Alpha Helix Pitch and Rise20m
- Alpha Helix Hydrogen Bonding24m
- Alpha Helix Disruption23m
- Beta Strand12m
- Beta Sheet12m
- Antiparallel and Parallel Beta Sheets39m
- Beta Turns26m
- Tertiary Structure of Protein16m
- Protein Motifs and Domains23m
- Denaturation14m
- Anfinsen Experiment20m
- Protein Folding34m
- Chaperone Proteins19m
- Prions4m
- Quaternary Structure15m
- Simple Vs. Conjugated Proteins10m
- Fibrous and Globular Proteins11m
- 5. Protein Techniques14h 5m
- Protein Purification7m
- Protein Extraction5m
- Differential Centrifugation15m
- Salting Out18m
- Dialysis9m
- Column Chromatography11m
- Ion-Exchange Chromatography35m
- Anion-Exchange Chromatography38m
- Size Exclusion Chromatography28m
- Affinity Chromatography16m
- Specific Activity16m
- HPLC29m
- Spectrophotometry51m
- Native Gel Electrophoresis23m
- SDS-PAGE34m
- SDS-PAGE Strategies16m
- Isoelectric Focusing17m
- 2D-Electrophoresis23m
- Diagonal Electrophoresis29m
- Mass Spectrometry12m
- Mass Spectrum47m
- Tandem Mass Spectrometry16m
- Peptide Mass Fingerprinting16m
- Overview of Direct Protein Sequencing30m
- Amino Acid Hydrolysis10m
- FDNB26m
- Chemical Cleavage of Bonds29m
- Peptidases1h 6m
- Edman Degradation30m
- Edman Degradation Sequenator and Sequencing Data Analysis4m
- Edman Degradation Reaction Efficiency20m
- Ordering Cleaved Fragments21m
- Strategy for Ordering Cleaved Fragments58m
- Indirect Protein Sequencing Via Geneomic Analyses24m
- 6. Enzymes and Enzyme Kinetics13h 38m
- Enzymes24m
- Enzyme-Substrate Complex17m
- Lock and Key Vs. Induced Fit Models23m
- Optimal Enzyme Conditions9m
- Activation Energy24m
- Types of Enzymes41m
- Cofactor15m
- Catalysis19m
- Electrostatic and Metal Ion Catalysis11m
- Covalent Catalysis18m
- Reaction Rate10m
- Enzyme Kinetics24m
- Rate Constants and Rate Law35m
- Reaction Orders52m
- Rate Constant Units11m
- Initial Velocity31m
- Vmax Enzyme27m
- Km Enzyme42m
- Steady-State Conditions25m
- Michaelis-Menten Assumptions18m
- Michaelis-Menten Equation52m
- Lineweaver-Burk Plot43m
- Michaelis-Menten vs. Lineweaver-Burk Plots20m
- Shifting Lineweaver-Burk Plots37m
- Calculating Vmax40m
- Calculating Km31m
- Kcat46m
- Specificity Constant1h 1m
- 7. Enzyme Inhibition and Regulation 8h 42m
- Enzyme Inhibition13m
- Irreversible Inhibition12m
- Reversible Inhibition9m
- Inhibition Constant26m
- Degree of Inhibition15m
- Apparent Km and Vmax29m
- Inhibition Effects on Reaction Rate10m
- Competitive Inhibition52m
- Uncompetitive Inhibition33m
- Mixed Inhibition40m
- Noncompetitive Inhibition26m
- Recap of Reversible Inhibition37m
- Allosteric Regulation7m
- Allosteric Kinetics17m
- Allosteric Enzyme Conformations33m
- Allosteric Effectors18m
- Concerted (MWC) Model25m
- Sequential (KNF) Model20m
- Negative Feedback13m
- Positive Feedback15m
- Post Translational Modification14m
- Ubiquitination19m
- Phosphorylation16m
- Zymogens13m
- 8. Protein Function 9h 41m
- Introduction to Protein-Ligand Interactions15m
- Protein-Ligand Equilibrium Constants22m
- Protein-Ligand Fractional Saturation32m
- Myoglobin vs. Hemoglobin27m
- Heme Prosthetic Group31m
- Hemoglobin Cooperativity23m
- Hill Equation21m
- Hill Plot42m
- Hemoglobin Binding in Tissues & Lungs31m
- Hemoglobin Carbonation & Protonation19m
- Bohr Effect23m
- BPG Regulation of Hemoglobin24m
- Fetal Hemoglobin6m
- Sickle Cell Anemia24m
- Chymotrypsin18m
- Chymotrypsin's Catalytic Mechanism38m
- Glycogen Phosphorylase21m
- Liver vs Muscle Glycogen Phosphorylase21m
- Antibody35m
- ELISA15m
- Motor Proteins14m
- Skeletal Muscle Anatomy22m
- Skeletal Muscle Contraction45m
- 9. Carbohydrates7h 49m
- Carbohydrates19m
- Monosaccharides15m
- Stereochemistry of Monosaccharides33m
- Monosaccharide Configurations32m
- Cyclic Monosaccharides20m
- Hemiacetal vs. Hemiketal19m
- Anomer14m
- Mutarotation13m
- Pyranose Conformations23m
- Common Monosaccharides33m
- Derivatives of Monosaccharides21m
- Reducing Sugars21m
- Reducing Sugars Tests19m
- Glycosidic Bond48m
- Disaccharides40m
- Glycoconjugates12m
- Polysaccharide7m
- Cellulose7m
- Chitin8m
- Peptidoglycan12m
- Starch13m
- Glycogen14m
- Lectins16m
- 10. Lipids5h 49m
- Lipids15m
- Fatty Acids30m
- Fatty Acid Nomenclature11m
- Omega-3 Fatty Acids12m
- Triacylglycerols11m
- Glycerophospholipids24m
- Sphingolipids13m
- Sphingophospholipids8m
- Sphingoglycolipids12m
- Sphingolipid Recap22m
- Waxes5m
- Eicosanoids19m
- Isoprenoids9m
- Steroids14m
- Steroid Hormones11m
- Lipid Vitamins19m
- Comprehensive Final Lipid Map13m
- Biological Membranes16m
- Physical Properties of Biological Membranes18m
- Types of Membrane Proteins8m
- Integral Membrane Proteins16m
- Peripheral Membrane Proteins12m
- Lipid-Linked Membrane Proteins21m
- 11. Biological Membranes and Transport 6h 37m
- Biological Membrane Transport21m
- Passive vs. Active Transport18m
- Passive Membrane Transport21m
- Facilitated Diffusion8m
- Erythrocyte Facilitated Transporter Models30m
- Membrane Transport of Ions29m
- Primary Active Membrane Transport15m
- Sodium-Potassium Ion Pump20m
- SERCA: Calcium Ion Pump10m
- ABC Transporters12m
- Secondary Active Membrane Transport12m
- Glucose Active Symporter Model19m
- Endocytosis & Exocytosis18m
- Neurotransmitter Release23m
- Summary of Membrane Transport21m
- Thermodynamics of Membrane Diffusion: Uncharged Molecule51m
- Thermodynamics of Membrane Diffusion: Charged Ion1h 1m
- 12. Biosignaling9h 45m
- Introduction to Biosignaling44m
- G protein-Coupled Receptors32m
- Stimulatory Adenylate Cyclase GPCR Signaling42m
- cAMP & PKA28m
- Inhibitory Adenylate Cyclase GPCR Signaling29m
- Drugs & Toxins Affecting GPCR Signaling20m
- Recap of Adenylate Cyclase GPCR Signaling5m
- Phosphoinositide GPCR Signaling58m
- PSP Secondary Messengers & PKC27m
- Recap of Phosphoinositide Signaling7m
- Receptor Tyrosine Kinases26m
- Insulin28m
- Insulin Receptor23m
- Insulin Signaling on Glucose Metabolism57m
- Recap Of Insulin Signaling in Glucose Metabolism6m
- Insulin Signaling as a Growth Factor1h 1m
- Recap of Insulin Signaling As A Growth Factor9m
- Recap of Insulin Signaling1m
- Jak-Stat Signaling25m
- Lipid Hormone Signaling15m
- Summary of Biosignaling13m
- Signaling Defects & Cancer20m
- Review 1: Nucleic Acids, Lipids, & Membranes2h 47m
- Nucleic Acids 19m
- Nucleic Acids 211m
- Nucleic Acids 34m
- Nucleic Acids 44m
- DNA Sequencing 19m
- DNA Sequencing 211m
- Lipids 111m
- Lipids 24m
- Membrane Structure 110m
- Membrane Structure 29m
- Membrane Transport 18m
- Membrane Transport 24m
- Membrane Transport 36m
- Practice - Nucleic Acids 111m
- Practice - Nucleic Acids 23m
- Practice - Nucleic Acids 39m
- Lipids11m
- Practice - Membrane Structure 17m
- Practice - Membrane Structure 25m
- Practice - Membrane Transport 16m
- Practice - Membrane Transport 26m
- Review 2: Biosignaling, Glycolysis, Gluconeogenesis, & PP-Pathway3h 12m
- Biosignaling 19m
- Biosignaling 219m
- Biosignaling 311m
- Biosignaling 49m
- Glycolysis 17m
- Glycolysis 27m
- Glycolysis 38m
- Glycolysis 410m
- Fermentation6m
- Gluconeogenesis 18m
- Gluconeogenesis 27m
- Pentose Phosphate Pathway15m
- Practice - Biosignaling13m
- Practice - Bioenergetics 110m
- Practice - Bioenergetics 216m
- Practice - Glycolysis 111m
- Practice - Glycolysis 27m
- Practice - Gluconeogenesis5m
- Practice - Pentose Phosphate Path6m
- Review 3: Pyruvate & Fatty Acid Oxidation, Citric Acid Cycle, & Glycogen Metabolism2h 26m
- Pyruvate Oxidation9m
- Citric Acid Cycle 114m
- Citric Acid Cycle 27m
- Citric Acid Cycle 37m
- Citric Acid Cycle 411m
- Metabolic Regulation 18m
- Metabolic Regulation 213m
- Glycogen Metabolism 16m
- Glycogen Metabolism 28m
- Fatty Acid Oxidation 111m
- Fatty Acid Oxidation 28m
- Citric Acid Cycle Practice 17m
- Citric Acid Cycle Practice 26m
- Citric Acid Cycle Practice 32m
- Glucose and Glycogen Regulation Practice 14m
- Glucose and Glycogen Regulation Practice 26m
- Fatty Acid Oxidation Practice 14m
- Fatty Acid Oxidation Practice 27m
- Review 4: Amino Acid Oxidation, Oxidative Phosphorylation, & Photophosphorylation1h 48m
- Amino Acid Oxidation 15m
- Amino Acid Oxidation 211m
- Oxidative Phosphorylation 18m
- Oxidative Phosphorylation 210m
- Oxidative Phosphorylation 310m
- Oxidative Phosphorylation 47m
- Photophosphorylation 15m
- Photophosphorylation 29m
- Photophosphorylation 310m
- Practice: Amino Acid Oxidation 12m
- Practice: Amino Acid Oxidation 22m
- Practice: Oxidative Phosphorylation 15m
- Practice: Oxidative Phosphorylation 24m
- Practice: Oxidative Phosphorylation 35m
- Practice: Photophosphorylation 15m
- Practice: Photophosphorylation 21m
Chemical Bonds - Online Tutor, Practice Problems & Exam Prep
 Created using AI
Created using AIChemical bonds are categorized into intramolecular bonds, which exist within a molecule, and intermolecular bonds, which occur between different molecules. Intramolecular bonds include ionic bonds, where electrons are transferred between atoms with opposite charges, and covalent bonds, where electrons are shared. Covalent bonds can be polar or nonpolar, depending on the electronegativity difference. Intermolecular forces include hydrogen bonds, dipole-dipole interactions, and van der Waals forces, which arise from temporary dipoles. Understanding these concepts is crucial for grasping molecular interactions and properties.
Chemical Bonds Introduction
Video transcript
Ionic & Covalent Bonds
Video transcript
So you guys already know that ionic and covalent bonds are types of intramolecular bonds that occur within a molecule. And recall that ionic bonds are interactions between atoms that have opposite charges due to losing and gaining electrons, but not sharing electrons. And recall that covalent bonds occur when 2 atoms share a pair of electrons, and those electrons can either be shared equally or unequally. If the electrons are shared equally between 2 atoms, then this forms a nonpolar covalent bond, a nonpolar covalent bond. Whereas, if the electrons are shared unequally, then this forms a polar covalent bond. And what determines the polarity of a bond is actually the difference in electronegativity, electronegativity between 2 atoms.
And so, in our example below, we're going to consider the 3 types of bonds. In the left column, we have ionic bonds. So, we know that ionic bonds do not share electrons, so we can put no in here. And in fact, what they do is they transfer electrons. So there's a transfer of electrons, but no sharing. Now, covalent bonds, regardless of the type, we know that there is sharing of electrons, so we can put yes in here. Now, for polar covalent bonds, we know that there is unequal sharing of electrons, whereas for nonpolar covalent bonds, we know that there is equal sharing of electrons. Now, for the electronegativity of the atoms, in ionic bonds, the atoms differ greatly in electronegativity, and there's actually a transfer of electrons. Now, for polar covalent bonds, there's also a significant difference, and in nonpolar covalent bonds, the atoms have either the same electronegativity or a very, very similar electronegativity.
And so, for examples, recall that the classic example of an ionic bond is table salt or sodium chloride, where the sodium has one less electron and the chlorine has one additional electron than proton, and so that gives them their charges. And the opposite charges are what keep this molecule intact or allow them to create an ionic bond. Now over here for polar, we know that water molecules are polar molecules because they have an oxygen and hydrogen. Oxygens are super electronegative, whereas hydrogens are not very electronegative, and so oxygens are going to have a partial negative charge and the hydrogens are going to have a partial positive charge, and that will occur on both hydrogens. And, over here with this molecule, the carbon dioxide, there are also polar bonds. So, you can see the carbon here is going to have a partial positive charge, whereas the 2 oxygens are going to have partial negative charges. But because the polarity is going in opposite directions, so you have electron density being pulled in opposite directions, the overall molecule, even though it contains polar covalent bonds, the overall molecule is nonpolar. So for a molecule to be considered polar, you also have to consider its shape. And so this is a nonpolar molecule that contains polar bonds. And then for nonpolar covalent bonds, we have this hydrogen gas molecule where we have 2 hydrogen atoms that have the same electronegativity, and so that creates a nonpolar bond where they share electrons equally.
So in our next video, we're going to recap intermolecular bonds. So, I'll see you guys in that video.
Intermolecular Forces
Video transcript
Recall that non-covalent intermolecular forces include hydrogen bonds, dipole-dipole interactions, and van der Waals forces. Recall that hydrogen bonds are interactions that involve a hydrogen atom, as well as electronegative atoms such as nitrogen, oxygen, or fluorine. Now a dipole itself simply explains a shift in the electron density that results due to electronegativity differences between two atoms. So, if you have a dipole on one molecule where there's an electron density shift, and then you have another dipole on a different molecule, that can create a dipole-dipole interaction between the two molecules, and so, that's what we see here. Now, van der Waals forces are forces that exist between all molecules because they result from instantaneous dipoles that can exist at any moment in any molecule.
So, down below in our example, we're going to label each of these diagrams with the appropriate intermolecular force. And so here, with these carbonyl groups on these different molecules, notice that there is a dipole moment going in this direction towards the oxygen in both molecules to create a partial negative charge on the oxygens and a partial positive charge on the carbons. And so, there's an attraction or an interaction between the partial positive carbon here and the partial negative oxygen on separate molecules, and so that is our dipole-dipole interaction. Now, over here on the right, we have water molecules, which are also polar molecules and have their dipoles. And so, each of these applies to all of these water molecules. And so, there's going to be a specific type of dipole-dipole interaction called a hydrogen bond between the hydrogen atoms on one molecule and the oxygen atoms on a different molecule, and so that is what a hydrogen bond is. And so notice that a single water molecule here can form up to four hydrogen bonds. So here, we will put hydrogen bonds.
Now, for our last example, we have these two nonpolar molecules, these two nonpolar hydrogen gas molecules, and so, overall, they don't have any partial charges because they have a similar electronegativity and there's no shift in electron density. There are no dipoles. However, at any single instant, there could actually be an instantaneous dipole where one of these hydrogens could have a partial negative, and the other could have a partial positive charge. And so, this is where our van der Waals forces come into play, existing between all molecules. This is a good summary of our intermolecular forces, and I'll see you guys in our practice videos.
Which of the following is classified as a nonpolar molecule?
Identify the types of chemical bonds present in scenarios #1 & 2 below:

Problem Transcript
Here’s what students ask on this topic:
What is the difference between intramolecular and intermolecular bonds?
Intramolecular bonds are the forces that hold atoms together within a molecule. These include ionic bonds, where electrons are transferred between atoms with opposite charges, and covalent bonds, where electrons are shared between atoms. Covalent bonds can be further classified as polar or nonpolar based on the electronegativity difference between the atoms. Intermolecular bonds, on the other hand, are forces that occur between different molecules. These include hydrogen bonds, dipole-dipole interactions, and van der Waals forces. Understanding the distinction between these types of bonds is crucial for grasping molecular interactions and properties.
 Created using AI
Created using AIHow do ionic and covalent bonds differ?
Ionic bonds occur when electrons are transferred from one atom to another, resulting in the formation of ions with opposite charges that attract each other. This typically happens between atoms with a large difference in electronegativity. Covalent bonds, however, involve the sharing of electrons between atoms. If the electrons are shared equally, the bond is nonpolar covalent; if shared unequally, it is polar covalent. The type of bond formed depends on the difference in electronegativity between the atoms involved.
 Created using AI
Created using AIWhat are hydrogen bonds and how do they form?
Hydrogen bonds are a type of intermolecular force that occurs when a hydrogen atom, covalently bonded to a highly electronegative atom like nitrogen, oxygen, or fluorine, interacts with another electronegative atom. This creates a dipole-dipole interaction where the hydrogen atom has a partial positive charge and the electronegative atom has a partial negative charge. Hydrogen bonds are crucial in many biological processes, including the structure of DNA and the properties of water.
 Created using AI
Created using AIWhat are van der Waals forces?
Van der Waals forces are weak intermolecular forces that arise from temporary dipoles in molecules. These forces exist between all molecules, regardless of their polarity. They result from the momentary distribution of electron density that creates a temporary dipole, which induces a dipole in a neighboring molecule. Although weak individually, van der Waals forces can collectively have a significant effect on the physical properties of substances, such as boiling and melting points.
 Created using AI
Created using AIHow does electronegativity affect bond polarity?
Electronegativity is a measure of an atom's ability to attract electrons in a chemical bond. When two atoms with different electronegativities form a bond, the electrons are not shared equally, resulting in a polar covalent bond. The atom with higher electronegativity will have a partial negative charge, while the other atom will have a partial positive charge. If the electronegativity difference is very large, the bond may become ionic. In contrast, if the electronegativities are similar, the bond will be nonpolar covalent, with equal sharing of electrons.
 Created using AI
Created using AI