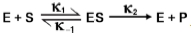Alright. So up until this point in our course, we've only briefly mentioned in some of our previous lesson videos, Michaelis-Menten, Michaelis-Menten Kinetics, or Michaelis-Menten equation. And so in this video, we're finally going to talk directly about Michaelis and Menten. We're going to introduce Michaelis and Menten and begin our discussion on the Michaelis-Menten enzyme kinetics assumptions. Michaelis and Menten are the last names of people. They were enzymologists or scientists that study enzymes. Michaelis was the last name of a German man named Leonor Michaelis. Below, we have an image of Leonor Michaelis, and we can see he was a pretty sharp guy with his perfectly groomed mustache and these super stylish antique glasses. Menten was the last name of a Canadian woman named Maud Menten. Below, we have an image of Maud Menten. Michaelis and Menten proposed a fundamental model to explain enzyme kinetics way back in 1913. What Michaelis and Menten proposed was that during an enzyme catalyzed reaction, it is necessary for an enzyme-substrate complex to form during the reaction. Before Maud Menten and Leonor Michaelis’ model, some people believed that enzymes would speed up chemical reactions by secreting some kind of chemical to speed up the reaction. We can see how Michaelis and Menten's model was incredibly important to enzyme kinetics. Recall that Maud Menten was a Canadian woman, and her accomplishments and her contributions to enzyme kinetics are impressively notable, especially considering that back in 1900, during her time in Canada, Canadian law did not give women the right to vote. Although controversial, some believed that the language included in Canadian law suggested that only a man could be considered a person. Technically, we could say that the Canadian law stated that women were not even considered people. It's incredibly impressive that Maud Menten was able to not only become a medical doctor but also get her Ph.D. and contribute so much to enzyme kinetics. Below what we have is the Michaelis-Menten equation. However, this is not the main focus of this video, and we will talk more about the Michaelis-Menten equation later in our course. For now, all I want you guys to know is that the Michaelis-Menten Enzyme Kinetics Model or equation will only work under a few simple assumptions. In our next lesson video, before we actually dive into detail on the Michaelis-Menten equation, we're going to talk about these particular assumptions that are needed for this Michaelis-Menten equation later in our course. That concludes our introduction to Michaelis and Menten and the Michaelis-Menten enzyme kinetics assumptions. I’ll see you guys in our next video where we'll discuss these assumptions in more detail.
Michaelis-Menten Assumptions - Online Tutor, Practice Problems & Exam Prep
Michaelis-Menten Assumptions
Video transcript
Michaelis-Menten Assumptions
Video transcript
So in our last lesson video, we said that the Michaelis-Menten equation, which we're going to talk more about later in our course, only works under a few simple assumptions. And so those assumptions can actually be condensed down into really three Michaelis-Menten assumptions. And so, again, the Michaelis-Menten equation that we'll talk about later in our course is only derived under these following three assumptions. And so notice that we've labeled our three assumptions with these three stars, and so these stars should look kind of familiar to you guys. And so do you guys remember in some of our previous lesson videos, we had these stars, and I said to remember these stars because we're going to talk about them more later in our course? Well, this is that video where we're going to talk about all three of those stars from our previous lesson video. And so the best part about this video and these three assumptions is that we've already covered all three of these assumptions in our previous lesson videos, and so really there's no new information in this video.
And so our first star here, or our first assumption, is the substrate concentration assumption, and that basically says to assume that the total substrate concentration substrate concentration is approximately equal to the free substrate concentration. And so in the Michaelis-Menten equation, we can use the free substrate concentration symbol S to represent the total substrate concentration. And so recall from our previous lesson videos, the reason that this is true is that the free substrate concentration is so much greater than the total enzyme concentration, which means that the concentration of the enzyme-substrate complex is negligible in comparison to the free substrate concentration. And so if we notice that the star number 1, we have an image for star number 1, and it's the same image that we used in our previous lesson video. So we already know that the substrate concentration is going to be much, much greater than the enzyme concentration. And because that's true, we can say that this symbol here, the free substrate concentration, can be used to represent the total substrate concentration. So moving forward, we're only going to see this symbol being used in our Michaelis-Menten equation, and that's all this first assumption is saying.
Now, moving on to our second star or our second assumption, it is the initial velocity assumption. And so the initial velocity assumption says that we're only going to focus on measuring the initial velocity of an enzyme-catalyzed reaction. And we already knew that. Right? Biochemists like to focus on the initial velocity of an enzyme-catalyzed reaction, and that is because the reverse reaction from the product backward into the enzyme-substrate complex, essentially the rate constant k-2, is essentially negligible really early on in the reaction since there's really not a lot of product early on in the reaction. And so again, this is all review and so down below you can see that for our second assumption here, all it's saying is that we're going to be measuring the initial reaction velocity. And so, this is really the main focus of biochemists when they're trying to study enzyme-catalyzed reactions. And, we know that the reverse reaction here, essentially from the product backward into enzyme-substrate complex, that is controlled by k-2 is essentially ignored early on in the reaction. Instead, all we have are k1, k-1, and k2 to consider when we are considering the initial velocity.
And so moving on to our 3rd and final star or our 3rd assumption, it is the steady-state assumption which we covered in our previous lesson videos as well. So we know that this is just saying that the concentration of the enzyme-substrate complex will remain constant. It will remain constant. And because it remained, remains constant, that means that the rate of formation or the rate constant for the formation of the enzyme-substrate complex will equal the rate of the dissociation of the enzyme-substrate complex or the sum of these two dissociation rate constants. And so again, we've already covered the steady-state assumption in our previous lesson video, so this is all review. And so looking down below, notice that we have the same graph that we had from our previous lesson videos for this third star. And so we can see that we have, in the blue here, we have the pre-steady-state period, and then in the yellow, we have the steady-state period. In the steady-state period for the steady-state assumption, we just say that the concentration of enzyme-substrate complex in red here stays constant. And so really these are the three assumptions that we need to know for the Michaelis-Menten equation moving forward in our course. And because we already are familiar with these from our previous lesson videos, it should feel a little bit like a review. And so we'll be able to apply some of these concepts moving forward in our practice problems, so I'll see you guys there.
Which of the following options is not an assumption made in deriving the Michaelis-Menten equation?
Michaelis & Menten assumed that the initial reaction for an enzyme catalyzed reaction could be written as shown:
Using this, the rate/velocity of ES-complex breakdown can be expressed by: