In this video, we're going to talk about yet another type of post translational modification, which is phosphorylation. So as we mentioned, phosphorylation is just one of many different types of post translational modifications and it's actually a pretty simple idea. All phosphorylation is is the covalent attachment of a phosphate group. And so it turns out that phosphorylation is actually one of the most common means of enzyme regulation, and therefore, that makes phosphorylation one of the most important forms of enzyme regulation. And that's why it's worth taking the time to study phosphorylation. Now down below in our image, we're showing you guys the simple idea of phosphorylation, where we can take any protein or any enzyme and simply add a phosphate group to the protein covalently so that we end up getting this structure right here. And, ultimately, this addition of the phosphate group to the protein can again regulate the enzyme's activity. And so in some scenarios, phosphorylation, the addition of this phosphate group, will activate the protein and turn on the protein so that it can do its job. Whereas in other situations, phosphorylation, again, the covalent attachment of this phosphate group, will inactivate the protein and turn off the protein so that it cannot perform its job. But, in either scenario, what's important to note is that phosphorylation is capable of changing the protein confirmation. So notice here we have a circular red protein and then over here in blue, we have the same exact protein just in a different confirmation, through this phosphorylation, the covalent attachment of this phosphate group. And so, in our next lesson video, we're going to talk about the exact source of the phosphate groups in phosphorylation. And we'll continue to talk more about phosphorylation as we move forward in our course. So I'll see you guys in our next video.
Phosphorylation - Online Tutor, Practice Problems & Exam Prep
Phosphorylation
Video transcript
Phosphorylation
Video transcript
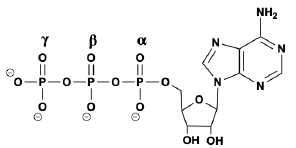
So now that we know that phosphorylation is just a post translational modification that involves the covalent attachment of a phosphate group, in this video we're going to talk about a common source of the phosphate groups in phosphorylation. And that is the high energy molecule, ATP, which I'm sure most of you guys are familiar with from your previous biology courses. And so recall that ATP is just an abbreviation for adenosine triphosphate. And the tri here means 3. So we know that adenosine triphosphate has threephosphategroups. And so that means that it has plenty of phosphate groups, and it doesn't mind donating a phosphate group for a phosphorylation event to occur. And so adenosine triphosphate or ATP molecules, as we'll see moving forward in our course, are typically going to be the source of the phosphate groups in phosphorylation. Now, when it comes to phosphorylation, really there are 2 major classes of enzymes that we need to consider. The first are going to be kinases, and the second are going to be phosphatases. And so kinases are going to be enzymes that catalyze phosphorylation reactions. Essentially, kinases are going to be the enzymes that add phosphate groups. Whereas, phosphatases are pretty much enzymes that catalyze the exact opposite reaction of kinases. And so what they do is they completely remove phosphate groups, from a substrate. And so if we take a look at our example image down below, notice on the left hand side, what we have is this red circle, which represents our un phosphorylated protein. So notice that there are no phosphate groups attached to it. And up above, we have this molecule here that has these threephosphategroups attached to it, and so this must be our ATP molecule. And so, again, kinases are going to be the enzymes that catalyze phosphorylation reactions. And so kinases are going to be able to use ATP as a co substrate and remove a phosphate group from the ATP and add it to the protein in order to get the phosphorylated protein that we have on the right over here. And so, notice that the phosphorylated protein has the attached the the covalently attached phosphate group. And, over here, what we have is the same molecule that we had over here on the left, except now it only has twophosphategroups instead of threephosphategroups. And so this makes it an ADP molecule, an adenosine diphosphate, and di, actually means 2. And so notice that the reverse reaction here, which essentially removes the phosphate group attached to the phosphorylated protein, is catalyzed by a phosphatase enzyme. And so really the main point of this lesson is to emphasize that ATP molecules are going to commonly be the source of the phosphate groups in phosphorylation events. And so, again, we'll see that a lot moving forward when we talk about different types of reactions, in metabolism. And in our next lesson video, we're going to talk about exactly which amino acid residues are most susceptible to phosphorylation. So I'll see you guys in that video.
During kinase phosphorylation, which phosphate group is removed from ATP in the figure below?
Phosphorylation
Video transcript
So now that we know that phosphorylation is a very common post translational modification that involves the covalent attachment of a phosphate group, and a common source of those phosphate groups is from ATP molecules. In this video, we're going to talk about which amino acids are most susceptible to phosphorylation. Actually, being phosphorylated makes a phosphoprotein. And so, all phosphoprotein is just a protein that has been phosphorylated. It's actually estimated that about one third of all of the proteins in a eukaryotic cell are phosphoproteins, which is a pretty big chunk of all those proteins, and it goes to show how important phosphorylation is as a form of regulation of proteins.
It turns out that amino acids that have hydroxyl groups are most susceptible to phosphorylation, and these hydroxyl groups in the R groups of these amino acids can be replaced with phosphate groups. Recall that hydroxyl groups are also called alcohol groups. We remember that even though alcohol is a serious threat, we can have it in moderation, and so a serious threat here reminds us that serine and threonine both have alcohol groups. Again, these hydroxyl groups and these amino acid R groups can be replaced with phosphate groups. It also turns out that histidine is occasionally phosphorylated as well, but it's more so phosphorylated in prokaryotic organisms like bacteria and in some plants, but not so much in human cells.
Phosphorylation or the covalent attachment of a phosphate group to these particular amino acids makes these amino acids even more polar than they already were, and that can ultimately lead to changes in the enzyme conformation or state. Essentially, changing the enzyme from the inactive T state to the inactive R state or vice versa. These phosphate groups that are attached through phosphorylation have an overall negative charge on them, and so that allows them to interact with positive charges on other groups through ionic interactions, and indeed, negative charges are going to repel other negative charges, and also these phosphate groups allow for stronger hydrogen bonding. These phosphate groups, having negative charges and allowing for stronger hydrogen bonding, are reasons for how phosphorylation can change an enzyme's confirmation or state.
And so, down below in our image, notice we're showing you the amino acids that can be phosphorylated. Over here on the left, notice that we have the same peptide backbone for all of these amino acids and we're focusing on the R groups here that can be phosphorylated. We know here what we have is a serine molecule but because the alcohol group is replaced with a phosphate group, this is actually a phosphoserine molecule. And here this one is going to be a phosphothreonine molecule. And over here, what we have is a phosphotyrosine molecule. And last but not least, over here on the far right, what we have is a phosphohistidine molecule. This green background right here can help remind you guys that phosphohistidine is more likely to be taking place in plants as well as in bacteria, and not so much in humans. This concludes our introduction to how amino acids can be phosphorylated, and as we move forward in our course, we'll be able to get some practice utilizing these concepts. So, I'll see you guys in our next video.
Covalent modification of an enzyme usually involves phosphorylation / dephosphorylation of:
When the active site of an enzyme is phosphorylated on one of its catalytic amino acid residues, the overall _______________ charge of phosphate groups would _____________ the affinity for a polar, negatively charged substrate.