In this video, we're going to begin our discussion on monosaccharide configurations. And so, of course, in your previous organic chemistry courses, you guys learned how to determine the configurations of chiral carbons. And so you may already remember from your previous courses that configurations of monosaccharide chiral carbons can be designated using two different systems. The first system is the Cahn-Ingold-Prelog system, which uses the letter R and the letter S to designate the configuration. And the second system is Fischer's convention, which uses the letter D and the letter L to designate configuration. And so moving forward in our course, we're going to talk about each of these two systems in separate videos, starting with the Cahn-Ingold-Prelog system. So I'll see you guys in our next video.
Monosaccharide Configurations - Online Tutor, Practice Problems & Exam Prep
Monosaccharide Configurations
Video transcript
Monosaccharide Configurations
Video transcript
So in this video, we're going to refresh your guys' memories of how to use the Cahn-Ingold-Prelog system to designate absolute configurations to chiral carbons using the letters R and S. The Cahn-Ingold-Prelog System is really just a synonym for R-S configuration.
Let's take a look at our sugar down below, glyceraldehyde. The chiral carbon is this carbon atom right here in the middle. Notice that the carbon at the top is not chiral because it has a double bond, or 2 bonds leading to the same chemical group of an oxygen atom. And the carbon at the bottom is also not chiral because it has two bonds leading to the same chemical group of a hydrogen atom. The only reason that the chiral carbon in the center is chiral is because it has 4 distinct chemical groups attached to it.
When we're trying to determine the configuration of this chiral carbon using the Cahn-Ingold-Prelog system and R-S configuration, these are the 3 steps that we really need to consider. The first is to assign priorities number 1 through 4 to each of the chemical groups that are attached to the chiral carbon. These assignments are based on the atomic numbers of the atoms in each chemical group. Looking at these 4 distinct pathways connected to this chiral carbon, we have a hydroxyl group, this entire group here, then this group down here, and, last but not least, this group to the left.
The oxygen atom has the highest atomic number and is assigned priority number 1. The carbon atoms in the blue and green groups have equal priority until you consider the substituents attached to those carbons. The carbon atom at the top has a double bond to an oxygen, whereas the carbon at the bottom only has one single bond to an oxygen, so the double bond has a higher priority. This pathway gets priority number 2, and the carbon atom down here gets priority number 3. That leaves the hydrogen atom with priority number 4.
In the second step, we determine if the configurations of priorities 1, 2, and 3 (ignoring 4), are arranged in a clockwise R configuration or a counterclockwise S configuration. After analyzing the priorities from 1 to 2, 2 to 3, and 3 back to 1, we see that it forms a counterclockwise pattern, which appears to be an S configuration.
The last and final step (step number 3) is only necessary if priority number 4 is on a wedge popping out of the page. Checking priority number 4, we see it's on a horizontal bond of the Fisher projection, which means it is indeed on a wedge. Therefore, even though this looks like an S configuration, because priority number 4 is on a wedge, this is actually an R configuration. So, this chiral carbon in the center is assigned an R configuration.
This video has outlined the steps we take to determine R-S configuration, which might be crucial as your professors could expect you to remember how to do this. Moving forward, we have some practice problems to help refresh your memories. Hopefully, this was a helpful refresher for you guys. In our next video, we'll talk about how to determine D-L configuration using Fisher's convention, so I'll see you guys there.
Monosaccharide Configurations
Video transcript
So now that we've refreshed our memories on how to designate RS configuration using the Cahn-Ingold-Prelog system, in this video, we're going to talk about how to designate DL configurations using Fisher's convention, which is actually more commonly used on carbohydrates since Fisher's convention was developed using carbohydrates. And so, Fisher's convention again is going to designate either D or L configurations relative to Glyceraldehyde enantiomers. And so, glyceraldehyde is just the name of one of the smallest and simplest sugars that exist. And again, enantiomers are just referring to non-superimposable mirror images.
And so, if we take a look down below at the image here in this box, notice that we're showing you the 2 Glyceraldehyde enantiomers. And so what's important to know is that the D configuration is going to be assigned to a chiral carbon when the hydroxyl group of that chiral carbon is pointing to the right, and the L configuration will be assigned to a chiral carbon when the hydroxyl group of that chiral carbon is pointing to the left of the Fischer projection. And so you can think L is for left. And so if we take a look down below at our Glyceraldehyde enantiomers, notice that this enantiomer over here has its hydroxyl group on its chiral carbon pointing to the right of the Fischer projection. And of course, if the hydroxyl group is pointing to the right, then we must assign a D configuration. And so that means that this glyceraldehyde enantiomer here is the D-Glyceraldehyde form and has D configuration.
Now notice looking at this enantiomer over here, the mirror image, its hydroxyl group on its chiral carbon is pointing to the left of the Fischer projection. And of course, a hydroxyl group pointing to the left corresponds with L configuration. And so this glyceraldehyde enantiomer over here is indeed the L-Glyceraldehyde form and has L configuration.
Now, one thing that's important to note here is that Fisher's convention will only properly work with standard Fisher projections. And recall that way back in our previous lesson videos, we covered standard Fisher projections. And so recall that standard Fisher projections will always have the carbonyl group on the top of the Fischer projection or towards the top of the Fischer projection. And so notice that with these glyceraldehyde enantiomers, the carbonyl group is hidden within the aldehyde group here, and so the carbonyl group is indeed on the top. And also recall that with standard Fisher projections, the longest carbon chain must be vertical. And so when we take a look at these Glyceraldehyde enantiomer over here, notice that the longest carbon chain is indeed vertical in both of these. So these are standard Fischer projections that are being shown. And most of the time, you will be given Fisher projections that are already in standard Fisher projection form, so that makes your job a little bit easier.
Now, usually under most circumstances that you'll encounter in your biochemistry course, D configurations are going to equal R configurations, and usually, L configurations are going to equal S configurations. But this is not always going to be true under every single circumstance. Again, this is going to be usually true under most circumstances that we will encounter. But if you want to be 100% safe, then all you really need to do is determine RS configuration separately from DL configuration.
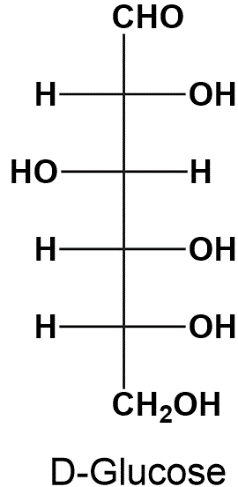
Alright. So now that we know how to determine DL configurations, let's go ahead and determine the DL configurations for all of the chiral carbons in these 2 glucose enantiomers shown over here. And so if we take a look at this first chiral carbon right here, notice that its hydroxyl group is pointing to the right. And of course, an OH group pointing to the right corresponds with D configuration. And so that means that we can give this chiral carbon right here a D configuration. So we'll put a little D right next to it. Now moving on to the next chiral carbon right here, notice that its hydroxyl group is pointing to the left. And of course, a left-pointing hydroxyl group corresponds with L configuration. So we can go ahead and give this chiral carbon right here an L configuration.
And then notice that these next two chiral carbons both have their hydroxyl groups pointing to the right, which again corresponds with D configuration. So we can go ahead and assign both of these chiral carbons D configuration. So now moving on to the next glucose enantiomer over here, notice that its first chiral carbon right here has its hydroxyl group pointing to the left, which of course corresponds with L configuration. So we can go ahead and give this chiral carbon an L configuration. Notice that the next chiral carbon right here has its hydroxyl group pointing to the right, so it's going to have a D configuration, so we can put D over here. And then the next 2 chiral carbons have both of their hydroxyl groups, or have their hydroxyl groups pointing to the left, which corresponds with L configuration. So we can put 2 Ls here.
And so the real question is, how do we determine the configuration of an entire monosaccharide if it contains a mixture of configurations, a mixture of D and L configurations. Well, really, that's what these next two bullet points are addressing right here. And so a monosaccharide will have the same exact configuration as the configuration of the highest numbered chiral carbon, or in other words, to say the same exact thing, we can say that a monosaccharide's configuration is going to be that of the chiral carbon that is furthest from its carbonyl group. And so if we take a look at this glucose enantiomer right here, notice that its carbonyl group is way up here at the top in this aldehyde group. And so the chiral carbon that is furthest away from the carbonyl group is going to be this chiral carbon right down here. And so that means that the configuration of this entire monosaccharide is going to be the same as this chiral carbon that's furthest away, which is a D configuration. And so that means that this entire glucose enantiomer is the D-glucose enantiomer.
Now if we take a look at this other enantiomer over here, notice that its carbonyl group, again, is at the very top, here in this aldehyde group. And the chiral carbon that's furthest from this carbonyl group is this one right here. And so this one has an L configuration, which means that this entire monosaccharide here will also have the L configuration, so this will be L-glucose. And so, really, this, you'll be able to get a good hang of, how this works as you start to get more practice. So moving forward in our course, we will definitely be able to apply these concepts in practice problems.
Now, the last thing that I want to leave you guys off with is that unlike biological amino acids, which we know from way back in our previous lesson videos are usually in the L configuration, biological carbohydrates on the other hand, they tend to usually be in the D configuration. And so, the real reason for why life prefers L amino acids but it prefers D carbohydrates is unknown. And so there are some theories, but we are not going to cover those theories here. But what you do need to know is that life prefers D carbohydrates and life prefers L amino acids. And of course, there are some exceptions, but for the most part, these are the preferences. And so this concludes our lesson on Fisher's projection and how to determine DL configuration. And again, we'll be able to get some practice as we move forward in our course. So I'll see you guys in our next video.
Identify the D/L and R/S configuration of all of the following monosaccharides:

Problem Transcript
How many D-stereoisomers are possible for a D-ketopentose?
Which TWO of the following traits apply to Fischer's Convention for naming stereoisomers?
a) Utilizes (+) and (-) designations.
b) Utilizes (D) and (L) designations.
c) Utilizes (R) and (S) designations.
d) Requires drawing the compound with aldehyde/ketone at the top.
e) Based on optical properties of the compound in solution.
f) Reflects stereochemistry at multiple centers.
Problem Transcript
How many possible epimers of D-glucose exist?