In this video, we're going to build up the knowledge to understand hemiacetals and hemiketals. Now before we actually define hemiacetal and hemiketal, it's first helpful to understand the reaction that results in cyclization of monosaccharides, and so monosaccharides, or sugars, can cyclize via a nucleophilic addition reaction that occurs between alcohol groups and the carbonyl groups of aldehydes or ketones. And so the alcohols in this reaction are going to be nucleophilic and so they will act as nucleophiles. Whereas the carbonyl groups of the aldehydes and ketones are electrophilic. So they are going to act as the electrophiles. And of course, the anomeric carbon is going to form upon cyclization of the monosaccharide. And so if we take a look at our example, we are going to see how alcohols can react with both aldehydes and ketones in order for a monosaccharide to cyclize. And so we can first analyze our image over here on the left and notice at the top here, we're just showing you this nucleophilic addition reaction that occurs between the alcohol and aldehyde. So notice here in orange, what we have is the alcohol group. And then, of course, over here in green, notice what we have is the aldehyde group. And so of course, the alcohol is going to be nucleophilic, so its electron density is going to attack the electrophilic carbonyl group of the aldehyde. And so of course, we're not showing the full mechanism that results upon monosaccharide cyclization, but this is the general idea that you guys should walk away with. Alcohols are nucleophilic, aldehydes are electrophilic. And so down below, notice that we're showing you a linear monosaccharide. And you can see that this linear monosaccharide has an aldehyde group and it has the alcohol group here, that is, needed for this nucleophilic addition reaction to take place. And so this is going to be an internal reaction that occurs within the same molecule. And ultimately, this results in this pyranose that we see over here when the C5 alcohol group interacts with the carbonyl group on the C1 carbon. And so this is a pyranose again because it contains a 6-membered ring. And notice that upon cyclization of this monosaccharide, the anomeric carbon forms. And of course, the anomeric carbon, we know from our previous lesson videos is the only carbon atom that's attached directly to 2 oxygen atoms, the ring oxygen and the oxygen of one of its alcohol groups. Now what you'll also notice is that the anomeric carbon before cyclization used to be the carbon of the carbonyl group. And so really that's another way to think about the anomeric carbon, it's the carbon atom that used to be part of the carbonyl group. Now, over here in the right side box, notice that we have a very similar reaction, except this time we have the alcohol group reacting with a ketone group instead of an aldehyde. And so again, the only difference between the aldehyde and ketone is that the ketone has the R group whereas the aldehyde has the Hydrogen. And so when we take a look at our linear monosaccharide here, notice that it contains both the ketone group and it contains the alcohol group. And so, this reaction is able to take place internally within the same monosaccharide and that results in the cyclization of the monosaccharide to form this furanose. And recall it's a furanose because it's a 5-membered ring when the C5 hydroxyl group interacts with the C2 ketone group. And of course upon cyclization, the anomeric carbon forms and, the anomeric carbon you can see is bound to 2 oxygen atoms directly and it used to be the carbon that was part of the carbonyl group. And so now that we have a better understanding of the reaction, that results in cyclization of monosaccharides. In our next video, we can distinguish between Hemiacetal and Hemiketal. So, I'll see you guys in that video.
Hemiacetal vs. Hemiketal - Online Tutor, Practice Problems & Exam Prep
Hemiacetal vs. Hemiketal
Video transcript
Hemiacetal vs. Hemiketal
Video transcript
So now that we're familiar with the reaction that allows monosaccharides to cyclize, in this video we're going to focus on how cyclization will form either a hemiacetal or a hemiketal. And so we already know from our last lesson video that the anomeric carbon will form upon monosaccharide cyclization. But what you may not have realized is that this anomeric carbon that forms will actually become part of a relatively unstable hemiacetal or hemiketal. Now, the prefix hemi is a prefix that means half, and so literally, hemiacetal can be defined as half of an acetal group. And so this is going to result from the reaction that we talked about in our last lesson video, which was an alcohol reacting with an aldehyde group. And then, of course, a hemiketal can be defined as half of a ketal group. And this is going to result from again, the same reaction we talked about in our last lesson video, which is an alcohol group reacting with a ketone group.
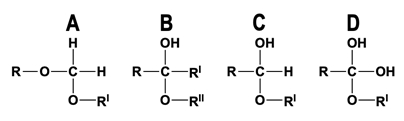
If we take a look at our example image down below, we can distinguish between hemiacetals and hemiketals. So in this box here, notice that the top half is showing the formation of a hemiacetal and the bottom half of our image is showing the formation of a hemiketal. And so what you'll notice is that the hemiacetal and hemiketal are resulting from the same exact reaction we talked about in our last lesson video, which is an alcohol group reacting with an aldehyde group will form this hemiacetyl and of course, an alcohol group reacting with a ketone group will form the hemiketal. Moreover, one thing that's interesting to note here is that the only difference between the aldehyde and the ketone is actually the difference that we see here, the hydrogen, and the R group. And really, when you take a look at it, the only difference between the hemiacetal and the hemiketal is the same exact hydrogen and R group. So notice that this hydrogen and this R group here is the only thing that makes the two different from each other.
So what you'll notice is that the carbon atom that we have highlighted here in the middle is the anomeric carbon. And again, the anomeric carbon is going to be part of either the hemiacetal or the hemiketal. And so, as we move forward through our course, we'll be able to easily distinguish them from each other. And really, it's just looking at the anomeric carbon and seeing if a hydrogen atom is attached or looking at the anomeric carbon and seeing if there's an R group attached. And so just to be clear here, you can think of these R groups as the rest of the carbon ring or the rest of the sugar structure, these R groups over here. Again, we're focusing on looking at these anomeric carbons here and just seeing if there is a hydrogen atom attached or if there is an R group attached.
So now that we've seen this, if we take a look at the previous image that we had up above from our previous lesson video, what you'll notice is that the anomeric carbon that we have here is a hemiacetal because notice that it has a hydrogen atom, just like the hemiacetal down here. And then notice over here on the right image, this is showing a hemiketal because going down, we have an R group, and so that is this R group that we have here. And again, as we move forward through our course, we'll be able to apply the concepts that we've learned here and you'll just get better and better at distinguishing between the Hemiacetal and the Hemiketal. So this concludes our lesson on the difference between a hemiacetal and a hemiketal, and I'll see you guys in our next video.
Which of the following is a hemiketal?
For monosaccharides to cyclize, an alcohol group must attack a carbonyl group within the same sugar.
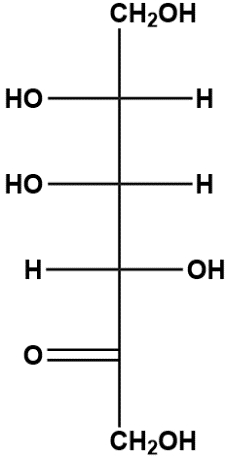
A) Which carbon of the linear ketohexose shown below has the reactive carbonyl?
a) C1.
b) C2.
c) C3.
d) C4.
e) C5.
B) Upon cyclization, would a hemiacetal or hemiketal form?
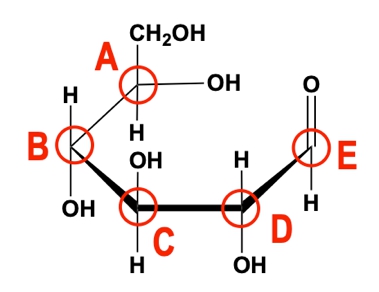
Use the image of the following glucose molecule to answer the questions.
A) Upon cyclization, which of the red circled carbons becomes the anomeric carbon?
a) A.
b) B.
c) C.
d) D.
e) E.
B) Upon cyclization, would a hemiacetal or hemiketal form?
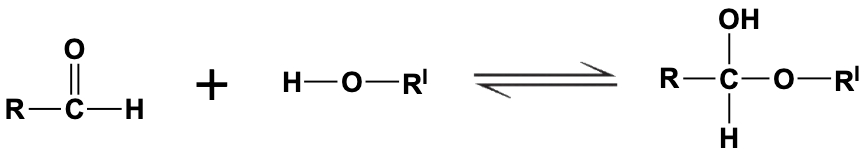
In the reaction shown below the compound on the far right is a: