In this video, we're going to introduce cyclic monosaccharides. Now, before we get started, it's important to note that up until this point in our course, we've mainly been focusing on the open or linear forms of monosaccharides. However, in biological solutions, most carbohydrates that have at least 5 carbon atoms exist in their cyclic forms, and so cyclic monosaccharides are very important when it comes to biochemistry. Now monosaccharides can actually cyclize to form many different types of rings, including 3, 4, 5, 6, and sometimes even 7-membered rings. However, the most common rings are 5 and 6-membered rings, and that's because 5 and 6-membered rings are actually the most stable rings. Now monosaccharides that have a 5-membered ring are called furanoses, and monosaccharides that have a 6-membered ring are called pyranoses. Now, of course, we know that the ending ose indicates a sugar, but why the prefix furan and why the prefix pyran? Well, it's because furanoses resemble the structure of furan. And so if we take a look at our image down below over here on the left-hand side, you can see that we're showing you the furan ring and the pyran ring structure down below. And so you can see that the furan ring is indeed a 5-membered ring. And so you can think the f in furan is for the 5-membered ring. And then, of course, the pyran ring is going to be a 6-membered ring. Now one thing to not get tripped up on is that the furan ring, even though it is a 5-membered ring, it does not have 5 carbons in its ring. Notice that it only has 4 carbon atoms in its ring, and the 5th member is actually an oxygen atom. And the same thing applies for the pyran ring. It is a 6-membered ring but does not have 6 carbons in its ring. Instead, it only has 5 carbons in its ring, and the 6th member is an oxygen atom. And the same thing applies to furanoses and pyranoses. And so notice over here, we're showing you the formation of a furanose. And down below, we're showing the formation of a pyranose. And so you can see that we're taking up at the top here the linear form of D-fructose. And then we've got this muscle man here bending the D-glucose molecule and really the muscle man is just here to help you guys better visualize the bending of the molecule. But the muscle man does not represent an enzyme as we'll find out later in our course. These cyclic structures will actually form without an enzyme. And so, here we can see that the C_5 hydroxyl group is going to interact with the ketone here on the C_2 carbon. And we'll talk more about this detailed reaction a little later in our course, but these two groups are going to react to create this furanose over here. And so you can see that the 5-membered ring resembles the 5-membered furanose ring, even though the double bonds aren't present over here, that's okay. You can still see the resemblance and that's why we call this D-fructofuranose. Now, moving on to the image down below, notice that we're starting with the linear form of the sugar D-glucose. And again, we've got this muscle man here just to help you guys better visualize the bending of the molecule and the muscle man does not represent an enzyme. However, here, notice that the C_5 hydroxyl group is interacting with the aldehyde group on the C_1 carbon instead of interacting with the ketone on the C_2. And so really that's the main difference that generates this pyranose over here. And again, you can see that this 6-membered ring resembles the 6-membered ring of the pyran even though the double bonds here and here are not present, over here. That's okay because we can still see the resemblance. And so this is going to be D-glucopyranose. And so, really, this here concludes our introduction to furanoses and pyranoses, and we'll continue to talk more about cyclic monosaccharides as we move forward in our course. So I'll see you guys in our next video
Cyclic Monosaccharides - Online Tutor, Practice Problems & Exam Prep
Cyclic Monosaccharides
Video transcript
Cyclic Monosaccharides
Video transcript
In this video, we're going to introduce a relatively easy way to convert Fisher projections into Haworth projections. And so all we need to remember is that chemical groups that are pointing to the left of a Fischer projection are going to be pointing upwards in a Haworth projection. And then of course, chemical groups that are pointing right of a Fisher projection are going to be pointing downwards in a Haworth projection. And so we can see over here with this Fisher projection, all of the left pointing groups are going to end up pointing upwards in the Haworth projection, whereas all of the right pointing groups are going to end up pointing downwards in the Haworth projection. If you remember the term uplifting, then you'll be able to associate left pointing groups of the Fisher projection with upwards pointing groups in the Haworth projection. And then if you remember the term downright, you'll be able to associate right pointing groups in the Fisher projection with downward pointing groups in the Haworth projection.
So let's go ahead and apply what we've learned here in our lesson to the D-glucose molecule that we see on the right. Notice that we have these very specific hydroxyl groups highlighted here, and we want to know if these hydroxyl groups are going to be pointing upwards or downwards in the Haworth projection. Looking at carbon number 2 right here, we can see that the hydroxyl group is pointing to the right. If it's pointing to the right, it's going to be pointing downwards in the Haworth projection. So with carbon number 2 here, we expect the hydroxyl group to be pointing downwards. So we can go ahead and put in an "OH" right here, and of course, this means that the hydrogen atom going over here is going to be pointing upwards since it's pointing to the left.
Now moving on to carbon number 3, notice that carbon number 3's hydroxyl group is pointing to the left. And of course, if you remember left, uplifting, you know that if it's pointing to the left, it's going to end up pointing upwards in the Haworth projection. So we know that the hydroxyl group is going to be pointing upwards. And, of course, that means that the hydrogen atom over here, which is pointing to the right, is going to be going downwards. So we can put the hydrogen here.
Last but not least, looking at carbon number 4, you can see that its hydroxyl group is also pointing to the right, which means that in the Haworth projection, it's going to be pointing downwards. And so, we can go ahead and put in the hydroxyl group going downwards, and of course, this means that the hydrogen atom, which is pointing left, is going to be pointing upwards in the Haworth projection.
By remembering uplifting, and downfall, you'll be able to easily convert Fisher projections into Haworth projections. That concludes this part of the lesson, and again, we'll be able to apply the concepts that we've learned here as we move forward in our course. I'll see you guys in our next video.
Cyclic Monosaccharides
Video transcript
So now that we've introduced cyclic monosaccharides like furanoses and pyranoses, in this video we're going to talk about how cyclic monosaccharide structures are commonly depicted with Haworth projections. And so if we take a look down below at our image, notice on the left-hand side over here, we have an open chain or a linear form of D-glucose. And of course, we know that this structure here is an example of a Fischer projection. Now over here, notice we have the cyclic form of D-glucose, shown as D-glucopyranose because it has a 6-membered ring. And so, what we're saying in this video is that cyclic monosaccharides are depicted with Haworth projection. So this is indeed a Haworth projection. And so what you'll notice is that in Haworth projections, these bonds towards the front are darker and thicker. And so the darker, thicker lines, you can imagine them as popping out of the page at us so that these bonds are closer to us as the readers. Also, notice that we have these lighter, thinner bonds that are in the back, and so these lighter, thinner lines, you can imagine them as going into the page, so that they are further away from us as the reader. And so really, the Haworth projection is supposed to bring a sense of 3-dimensionality, just like the Fischer projection is supposed to bring a sense of 3-dimensionality.
Now, also, just like there are standard Fischer projections where the carbonyl group is towards the top and the longest carbon chain is vertical, there are also standard Haworth projections. And so the standard Haworth projections will have what is known as the anomeric carbon, on the right side of the structure, and we'll define the anomeric carbon later in our course. However, the anomeric carbon in this structure is this carbon right here. So the anomeric carbon is on the right-hand side. And, with standard Haworth projections, the highest numbered carbon, so numbering these carbons, is going to be pointing upwards. And so you can see that the highest numbered carbon, these red numbers show, the numbering of the carbons, and you can see that the highest numbered carbon, which is carbon 6 here, is pointing in an upwards fashion. And so, really this is the standard Haworth projection. But again, we'll get more practice utilizing and recognizing standard Haworth projections as we move along through our course. But really, the main takeaway here in this lesson is that Haworth projections are used for cyclic monosaccharides whereas Fischer projections are used for linear monosaccharides.
Now, as we move forward, it's also important to keep in mind that Haworth projections can actually sometimes be a little bit misleading, and that's because cyclic monosaccharides are actually not planar. So when we look at this Haworth projection, you might assume that all of these carbons that are being shown here, all of the members of the ring, I should say, all of them, you might assume, are in the same plane because they appear to be flat in this Haworth projection. But that's a little bit misleading, as I said because the cyclic monosaccharide here is not planar, and that's because recall that every carbon atom is going to have tetrahedral geometry. And so later in our course, we're going to talk about other ways to display cyclic monosaccharides. And so this here concludes our introduction to Haworth projections and again we'll be able to apply these concepts as we move forward in our course. So I'll see you guys in our next video.
Cyclic Monosaccharides
Video transcript
So recall that in one of our older lesson videos, we had talked about how to assign numbers to the carbon atoms of linear monosaccharides. But in this video, we're going to talk about how to assign numbers to the carbon atoms of cyclic monosaccharides, which is actually going to be a little bit different. The carbon atoms in cyclic monosaccharides are going to be numbered based on the positioning of what's known as the anomeric carbon. But what in the world is the anomeric carbon? Well, the anomeric carbon is going to be the only carbon atom that is covalently attached to two oxygen atoms directly.
And so, if we take a look down below at our cyclic sugar molecule, notice that the anomeric carbon is highlighted with this green background right here. And so if we take a closer look at this anomeric carbon, notice that it is directly bound to two oxygen atoms. One oxygen atom is the ring oxygen, and the other oxygen atom is the oxygen of the hydroxyl group. If you take a look at all of the other carbon atoms, none of these carbon atoms are covalently attached to two oxygen atoms like the anomeric carbon is, and so that makes it pretty easy to identify the anomeric carbon.
Now, when it comes to numbering the carbon atoms of cyclic monosaccharides, we need to prioritize making the anomeric carbon the lowest possible number. We want to assign the anomeric carbon the lowest possible number. When we take a look at the anomeric carbon over here, the lowest possible number that we can give it is carbon number 1. And then, of course, we can start to number the carbons sequentially as they are connected. So this carbon atom would be carbon number 2. This carbon atom would be carbon number 3. This carbon atom is carbon number 4. This is carbon number 5 and then carbon atom number 6 is up here. This is the proper numbering of the carbon atoms in this glucose molecule in our example.
As we move forward in our course, we'll be able to get some practice identifying the anomeric carbon and we'll get some practice numbering the carbon atoms of cyclic monosaccharides. So I'll see you guys in our next video.
Below is the structure for a cyclic D-monosaccharide. Which labeled carbon is the anomeric carbon?
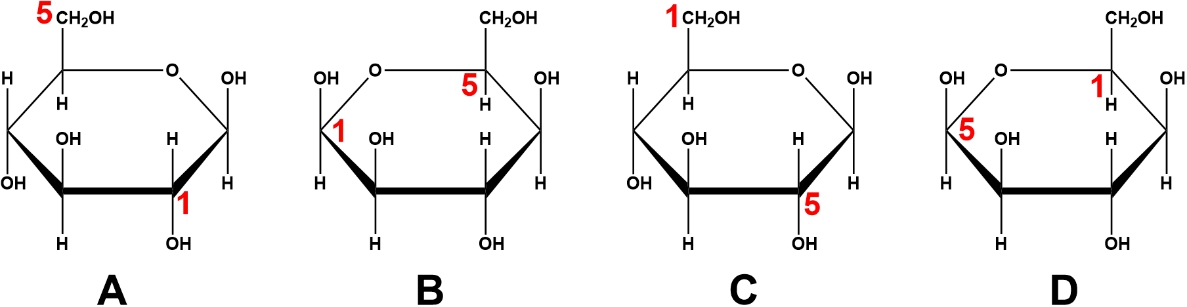
Which image represents the proper convention for carbon numbering of cyclic sugars?