In this video, we're going to begin our lesson on functional groups. Now, as their name implies, functional groups are really just groups of atoms that are reactive or functional, and they're also commonly found together within biomolecules. Moving forward in our course, when we discuss different classes of biomolecules, we're going to see lots and lots of functional groups appear, and it's important to get to know these functional groups pretty well. The functional groups typically extend off of the carbon backbone of a molecule. Notice below in our image, we represent the carbon backbones of the molecules by using these squiggly lines throughout, which again represent the carbon backbone of the rest of the molecule, and so the functional group is always going to be extending off of some type of carbon backbone.
Throughout all of biology, there are a lot of different types of functional groups. However, in a typical biology course like yours, you're likely only going to need to know 7 functional groups that are pretty common in biology. Notice below in our table, we have these 7 functional groups for you all, to know. The very first functional group that you all should know is the methyl group. The methyl group is just when you have a carbon atom branching off of a carbon backbone, and this carbon atom is covalently attached to 3 other hydrogen atoms. Typically, these methyl groups are going to be found in lipids and such.
The second functional group that you all should know is the hydroxyl group. The hydroxyl group, as you can see with its name, has the "oxy" in here for an oxygen atom and the "hydro" in here for a hydrogen atom, and that's exactly what it is. The hydroxyl group is going to have an oxygen atom branching off of a chain that's also bound to a hydrogen atom. We'll see hydroxyl groups in lots of different types of molecules including carbohydrates.
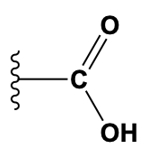
The third class of functional group is the carbonyl group. The carbonyl group occurs whenever you have a carbon atom that has a double bond to an oxygen atom. These are fairly common throughout different types of biomolecules. The fourth functional group is the carboxyl group. I admit, at first glance, the carbonyl and the carboxyl sound very similar. However, the carboxyl is a combination of the hydroxyl group and the carbonyl group as well. Notice that the carbonyl group is present here in the carboxyl because there's a carbon double bonded to an oxygen just like what we saw over here. But in addition to the carbonyl group, there's also a hydroxyl group over here within this carboxyl group.
The fifth functional group down below is the amino group. The amino group, as you can think, has this "n" in it, representing the nitrogen atom that is found within the amino groups. The sixth functional group is the phosphate group, and the phosphate group looks complex; however, it's recognizable because it's the only one of the 7 that has a phosphorus atom like what we see here. The seventh and final functional group that you all should be aware of is the sulfhydryl group. As its name implies, with the "sulf" here, it's going to have a sulfur atom, and the "hydra" prefix here is going to have a hydrogen atom. That's exactly what the sulfhydryl group is, a sulfur and a hydrogen atom.
These are the 7 functional groups that would be good to commit to memory because moving forward in our course, we're going to refer to all of these different functional groups. Some of them you might need to commit to memory in terms of structures, but others you might need to commit to memory just in terms of being able to identify or recognize them. For example, the phosphate group might be one that you would just need to identify and recognize. But you'll have to ask your professor to figure out exactly which functional groups they want you to be aware of. This concludes our introduction to functional groups, and we'll be able to get some practice applying the concepts that we've learned in our next few videos. I'll see you all there.